- Following EpiPen controversy, federal watchdog to review Medicaid rebates (statnews.com)
A government watchdog agency is conducting three new reviews of the Medicaid Drug Rebate Program to determine the extent to which drug companies are properly paying required givebacks to the health care program...Under the Medicaid Drug Rebate Program, companies must accurately report — and pay — a rebate on drugs paid for by the agency...The ruckus erupted because Mylan reported EpiPen as a generic product for nearly a decade. This is an important distinction, because classifications are used to determine the size of rebates...are lower for generics — 13 percent versus 23 percent for a brand-name product...The more we learn about the misclassifications, the more we know this program is in dire need of proper oversight...It’s unclear whether CMS took any action between 2009 and now to get the EpiPen classification back on track after being told about the misclassification...
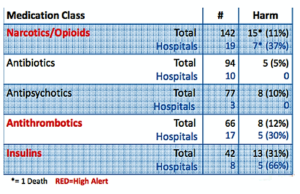
- ISMP Names Top Medication Safety Issues of 2016 (pharmacytimes.com)
It’s crucial for pharmacists to be aware of medications associated with high risk for error and harm to patients, and to look for best ways to implement practices for improving safety and patient care...The Institute for Safe Medication Practices...Compiled data gathered from hospital medication error reports, risk assessments, consumer reports, and FDA collaboration...to name the top medication classes involved in adverse events...the top 5 high-alert medication classes based on data from 2016. Opioids, antithrombotics, and insulins topped the list, followed by antipsychotics and antibiotics...data pegged wrong dosage as the top reason for adverse events in most cases, except in the use of antibiotics, for which wrong drug was the top reason...
- Makers of Abilify drug settle with 43 states, including Nevada, for $19.5M (reviewjournal.com)
Nevada is expected to receive nearly $300,000 in a multistate settlement with a pharmaceutical company accused of marketing a drug to patient groups for which they didn’t have federal approval and mischaracterizing the drug’s risks...Bristol-Myers Squibb Company, which partnered with Otsuka America Pharmaceutical, Inc. to market antipsychotic drug Abilify, settled with 43 states for $19.5 million, according to an announcement Thursday by the office of Nevada Attorney General Adam Laxalt...The company was accused of marketing the drug to seniors with Alzheimer’s disease and dementia despite the fact the drug wasn’t approved for those purposes by the U.S. Food and Drug Administration...
- U.S. Doctors Still Over-Prescribing Drugs: Survey (realclearhealth.com)
Despite evidence that certain drugs aren't always necessary, doctors are still prescribing these treatments, a new survey of doctors reveals...Antibiotics are by far the drugs most frequently used in situations where they'll provide no value for patients. The survey found that more than a quarter of doctors surveyed (27 percent) said that antibiotics are often administered to patients when the drugs will do no good...antibiotics are prescribed to treat upper respiratory infections even though these are most often caused by viruses unaffected by the medication…Other treatments that doctors use frequently despite their questionable value include aggressive treatments for terminally ill patients (9 percent), drugs prescribed for chronic pain (7 percent), and dietary supplements such fish oil and multivitamins (5 percent), the survey revealed...Physicians also might be practicing defensive medicine to ward off potential malpractice suits, or trying to meet clinical performance measures that call for blanket treatments that must be applied to all patients...
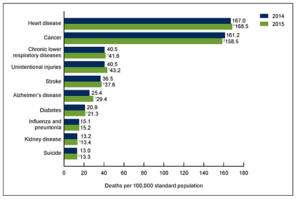
- 5 Findings From the Recent US Mortality Data Report (ajmc.com)The CDC’s National Center for Health Statistics released a report (Mortality in the United States, 2015) with new data on the 2015 US mortality rates. Here are 5 findings to take away from the report...the National Center for Health Statistics...released a data brief containing information on US death rates, life expectancy, and infant mortality rates in 2015. Here are 5 findings to take away from the report.
- In a troubling sign, life expectancy fell.
- The age-adjusted death rate has increased as well.
- The leading causes of death remain the same…
- … But rates of those causes have changed.
- Infant mortality has risen, but not significantly.
- Pharmacy Week in Review: December 9, 2016 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Five things for pharma marketers to know: Wednesday, December 7, 2016 (mmm-online.com)
- President-elect Donald Trump told Time magazine that he “doesn't like what's happened with drug prices” and that he plans to bring down the cost of prescription medications. Time named Trump its Person of the Year on Wednesday. (Time)
- Mylan plans to lay off nearly 10% of its global workforce. A Mylan spokesperson said the job cuts are part of an efficiency strategy. (BioSpace)
- Pfizer was fined $107 million for overcharging the U.K.'s health system for a generic epilepsy drug, phenytoin sodium. The country's antitrust regulator said that Pfizer deliberately unbranded the drug to skirt a competition law. Pfizer said it plans to appeal the ruling. (WSJ)
- Celgene said its understanding of how its multiple myeloma drug Revlimid works in cancer patients is a boon for its R&D efforts in other diseases. The drugmaker currently has three drugs in its pipeline: CC-122 for diffuse large B-cell lymphoma, CC-220 for lupus, and CC-90009 for acute myeloid leukemia. (Bloomberg)
- ICYMI: GlaxoSmithKline said the CEO of ViiV healthcare, Dr. Dominique Limet, will step down and that Deborah Waterhouse, the company's SVP of primary care at GSK U.S. pharmaceuticals, will succeed him. ViiV Healthcare is an HIV specialty company formed in 2009 by GSK and Pfizer.
- This Week in Managed Care: December 9, 2016 (ajmc.com)
Sara Belanger, with The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network.
- Map of all medicines and their mode of action is created (pharmaceutical-journal.com)
Scientists have mapped all 1,578 licensed drugs licensed by the US Food and Drug Administration according to their mechanisms of action to help researchers visualize the ‘uncharted waters’ where they may find future treatments...Scientists at The Institute of Cancer Research in London...extracted data from their own drug database, as well as databases at European Bioinformatics Institute in Cambridge and the University of New Mexico. They matched each drug with prescribing information and data from published scientific papers to build up a picture of how each existing medicine works. The analysis, published in Nature Reviews Drug Discovery (A comprehensive map of molecular drug targets) reveals that there are just 667 unique human proteins targeted by existing approved drugs (or only 3.5% of the estimated 20,000 human proteins), and a further 189 drug targets in pathogenic organisms...This new map of drugs, created through the latest computational analytical technologies, will enhance our ability to use rational, data-driven approaches to identify the most promising future targets and treatment combinations for the next generation of cancer and other diseases...
- The Cost of Counterfeits (pharmtech.com)
The proliferation of counterfeit medicines is nothing new to pharma; however, the scale of the problem seems to be escalating, especially with the Internet providing an easy means for fraudsters to dispense their fakes. Counterfeiting has a devastating impact on public health and the economy. Not only are consumers paying for products of inferior quality, but their well-being is also put at risk. For genuine drugmakers, profits are diluted, but the repercussions extend beyond that...The European Union Intellectual Property Office reported...that the pharmaceutical industry is stripped of approximately €10 billion of revenue every year because of counterfeit medicines; this figure accounts for 4.4% of the sector’s sales...the lost sales translate into 37,700 jobs lost across the pharmaceutical sector in the EU as a result of legitimate manufacturers and distributors employing fewer people than they would do had this problem not existed…With serialization and track-and trace legislations being rolled out over the next few years, pharma is doing its part to secure its supply chain. The problem will be an ongoing challenge for the industry, but with advances in technology, it will become easier to detect the fakes in the near future.