- What Services Should Patients Expect from a Specialty Pharmacy? (specialtypharmacytimes.com)
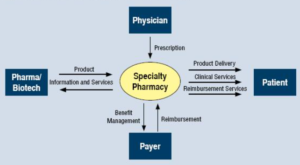
There are 2 important stakeholders when it comes to specialty medications: the patients and the providers. Both groups have a vested interest in the clinical outcome..."what would I expect if I were a specialty patient or a specialized provider?"...Once the reality of the condition begins to set in, then the needed medication must be started to improve clinical outcomes. How would the ideal specialty pharmacy interact with both the patient and their provider?...The ideal specialty pharmacy would have clear upfront communication with both parties. Proactive communication and referral management for the provider would allow the specialist to know precisely what services and capabilities the specialty pharmacy provides...The provider then could inform the newly diagnosed patient that...their expertise, and that of the specialty pharmacy, will ensure the best possible clinical outcomes. At this point, the referral has been sent to the specialty pharmacy, and the patient's interaction with the specialty pharmacy begins.
- Dept of Health to launch sepsis awareness campaign across UK (pmlive.com)
An awareness campaign to improve public recognition of sepsis will be launched across the UK this year, carried out by the Department of Health and Public Health England...Working in collaboration with the UK Sepsis Trust, the initiative aims to better public understanding of the blood poisoning condition that requires quick diagnosis and treatment to effectively combat...Approximately 150,000 adults and children in the UK are affected by sepsis annually, with around 44,000 of those dying each year. If not identified and treated quickly, survivors of sepsis can be left with life-changing disabilities... better sepsis care could save 13,500 lives every year and save the NHS £314m annually...The public awareness drive - called Just ASK: could it be sepsis? - hopes to equip the general public with knowledge of the condition so that they might recognise it and prompt healthcare professionals to fast-track diagnosis of the time-sensitive illness.
- Device makes single doses of drugs on demand (upi.com)Synthetic biology and microbioreactor platforms for programmable production of biologics at the point-of-care (nature.com)
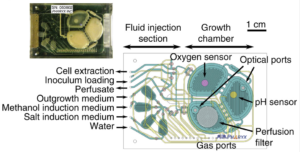
A portable device may allow doctors to create single doses of biopharmaceutical medications on demand, potentially speeding the treatment of diseases that include diabetes and cancer...The portable production system was developed by scientists at the Massachusetts Institute of Technology, with funding from the U.S. Defense Advanced Research Projects Agency, expecting the device could be useful for the battlefield and remote areas to produce treatments immediately at the point of care...The system...can currently produce two biologic drugs from a single yeast strain in the device, creating near-single-dose production in less than 24 hours with limited infrastructure...The production system uses a programmable strain of yeast, Pichia pastoris, which produces two proteins used for disease therapy...The strain of yeast, which can grow at very high densities when exposed to carbon sources, expressed recombinant human growth hormone when exposed to estrogen β-estradiol and expressed the protein interferon when exposed to methanol...The potential use for the device is significant, as it can be used for everything from treatments on a battlefield where immediate care is required to prevention of a disease outbreak in a remote village...
- Nevada State Board of Pharmacy Newsletter – July 2016 (bop.nv.gov)
- Must I Get an ID on My Patient? - ChaoFen (Stacy) Tan, PharmD Candidate
- Prescription Over-the-Counter Products
- FDA Calls for Review of Opioids Policy, Announces Action Plan
- More Selected Medication Safety Risks to Manage in 2016 - ISMP
- Manufacturer Drug Labeling, Packaging, Nomenclature - Per Liter Electrolyte Content on Various Sizes of Manufacturers' IV Bags
- Patient Education - Discharging Patients Who Do Not Understand Their Discharge Medications
- Compliance News
- USP Publishes Chapter on Handling Hazardous Drugs in Health Care Setting
- FDA Provides Training Video on Keeping Medications Safe in Emergency Situations
- FDA Requires Class-Wide Labeling Changes for IR Opioid Analgesics
- FDA Issues Alert Regarding All Unexpired Sterile Drug Products Produced by Medaus Pharmacy
- E-Prescribing - Who Can Send Electronic Prescriptions? - Melissa Hampton, PharmD Candidate
- Veeva Streamlines Drug Giants’ Ability To Get Medicine Approvals (investors.com)
When health care behemoth Johnson & Johnson decided last year to use Veeva Systems' software for gathering data from clinical trials needed to get a new drug approved, Veeva's rivals took notice..."They (J&J) said we're going to standardize this globally, and within 12 months we had 6 customers (among the 20 biggest drugmakers) do the exact same thing,"...one... goal in mind -- replace biopharma companies' legacy systems with cloud-based software designed to make their painstaking regulatory requirements a lot less painful...The cost of developing a new drug has soared to $2.6 billion, up from about $1 billion in 2000 in inflation-adjusted dollars, according to the Pharmaceutical Research and Manufacturers of America. Because the stakes are so high, more large drug developers have decided in rapid succession to junk their prior technology investments...The key to Veeva's success is that it has brought together enterprise software expertise with industry-specific expertise...
- Hep C Services Pharmacists Can Provide (pharmacytimes.com)
Jennifer Andres, PharmD, BCPS, clinical assistant professor of pharmacy practice at Temple University School of Pharmacy discusses the sorts of services pharmacists can provide for hepatitis C patients.
- ACO Growth Impact on Specialty Pharmacy (specialtypharmacytimes.com)
Kevin James, RPh, MBA, vice president of payer strategy at US Bioservices, discusses the impact of Accountable Care Organizations on specialty pharmacy.
- GSK and Google launch bioelectronics venture (pmlive.com)Q&A: Glaxo exec says bioelectronics is ‘not science fiction’ (statnews.com)
GlaxoSmithKline has joined forces with Google's Verily Life Sciences to establish a new company dedicated to the development of bioelectronic medicines...Galvani Bioelectronics - named for the 18th century Italian bioelectricity pioneer - will combine GSK's drug discovery and development expertise in disease biology with Verily's expertise in highly miniaturised technologies, including data analytics and software development for clinical application...Together, we can rapidly accelerate the pace of progress in this exciting field, to develop medicines that truly speak the electrical language of the body...Bioelectronic medicine is concerned with the electrical signals firing between the body's nervous system and organs, working to regulate the faulty nerve impulses that occur in many illnesses.
- Pfizer agrees to settle shareholder class action on Celebrex and Bextra (fiercepharma.com)
Pfizer’s Bextra (valdecoxib) has not been on the market in more than a decade, but the litigation tied to it and pain drug Celebrex (celecoxib) continues to play out. The New York drugmaker has now put to rest a long-running class action by Pfizer shareholders who said a controversy over the drugs’ safety whacked the stock price and cost them a lot of money...Pfizer last week reached an agreement-in-principle to resolve the securities class action case for all defendants...the resolution is pending court approval and the terms are confidential until a formal agreement has been approved...The pain drugs made Pfizer a lot of money, at one time being among the company’s best-selling meds. But the controversy over their risks has also cost it tremendously. In 2009, Pfizer agreed to hand over $2.3 billion to settle a Department of Justice probe into its marketing of Bextra...And last year, the drugmaker settled another related investor suit for $400 million.
- Gold Stars for Stellar Adherence (pharmacytimes.com)
Medicare Advantage and other supplemental Part D plans encourage medication adherence to achieve higher star ratings... This rating system measures insurance plan quality, as higher quality plans improve patient outcomes and minimize costs. Adherence represents 10.9% and 29.5% of Medicare Advantage and supplementary plans’ star ratings, respectively...Community pharmacies...have initiated automatic refill programs to improve adherence. These programs have the added benefit of boosting insurance plans’ star ratings...(CMS) confidently assumes that patients of brick-and-mortar pharmacies use their refills if they pick up the prescriptions...researchers reported that an automated prescription refill program improved medication adherence of Medicare Part D patients...Easing barriers to refilling prescriptions enhances adherence, and reinforced adherence enhances pharmacy sales directly and indirectly. Insurance companies prefer pharmacies that strengthen their star ratings; they’re less likely to engage with pharmacies with mediocre adherent patient populations from coverage, and they provide bonuses to pharmacies for high adherence...Future studies assessing the impact of automatic refill programs should develop a superior method of measuring actual medication intake.