- This Week in Managed Care: July 9, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Painkiller panel drops experts linked to pharmaceutical industry (financialexpress.com)
One of the experts, Dr. Gregory Terman, said he was dismissed Tuesday afternoon by phone. He said he was told the decision was made because his nonprofit group, the American Pain Society, receives funding from drugmakers...A group advising the Food and Drug Administration on medical issues abruptly dropped four experts from a panel on prescription painkillers after concerns emerged about apparent ties to the pharmaceutical industry...Federal advisers are supposed to be vetted for financial ties that can influence their judgment. Senator Ron Wyden of Oregon sent a letter Friday to the academies’ leadership noting that two of the panel nominees had also served in professional societies that receive funding from drugmakers. Wyden has protested industry influence on federal expert panels before.
- IACP’s 2016 Compounders on Capitol Hill Report (iacprx.org)
IACP Members Go to the Hill with Three Actionable Asks
This year, IACP went to the Hill with three Asks, each with a Congressional action item. IACP and its legislative team have worked tirelessly for the past year emphasizing FDA should be implementing DQSA (Drug Quality and Security Act) according to congressional intent!
- Issue #1 Support Pharmacists: Sign the Bipartisan Letter to FDA Led by Representatives Chris Stewart and Henry Cuellar.
- Issue #2 Support Letter to Chairman Chaffetz and Ranking Member Cummings to Request House Oversight and Government Reform Hearings on FDA's Implementation of DQSA.
- Issue #3 Support Pharmacists: Sign the Senate Letter to FDA Regarding Office-use.
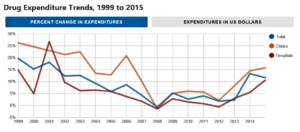
- Pharmacy Journal Examines National Trends in Prescription Drug Spending (ashp.org)National trends in prescription drug expenditures and projections for 2016 - Abstract (ajhp.org)
A sharp rise in prescription medication prices — driven by the introduction of new, expensive specialty medications and huge increases in the costs of older drug products with few competitors — drove an 11.7 percent increase in spending on medications in 2015, according to a new report published in AJHP (American Journal of Health-System Pharmacy)...The report projects an 11 to 13 percent increase in total drug expenditures in hospitals and health systems in 2016, which includes a 15 to 17 percent increase in clinic spending and a 10 to 12 percent increase in hospital spending...pharmacists need to be diligent about understanding what factors influence drug spending within their own organizations. "By understanding the key cost drivers, pharmacy leaders can implement programs to target them," he noted, adding that such strategies could include working with prescribers to use equally effective but less expensive medications or developing programs such as antibiotic stewardship to reduce unnecessary use of drugs...
- Viagra Price Drops 50% as Drug Faces Competition (247wallst.com)
Viagra, the wildly successful erectile dysfunction pill, faces challenges from a generic version that will launch next year, and direct competitors Levitra and Cialis. In the face of these, Viagra has started a promotion that offers 50% off a patient’s next three prescriptions...The offer does come with some fine print: home delivery ...In March, the mammoth generic drug company Teva Pharmaceutical Industries Ltd. got approval to market a generic version of Viagra. Teva already had FDA approval of its product. Due to a deal between Pfizer Inc. and Teva, the product will not come on market until the end of 2017. That leaves only 18 months for Pfizer to sell Viagra without generic competition. After that, it faces the Teva drug, which almost certainly will have a lower price.
- Infection experts warn of more U.S. superbug cases in coming months (reuters.com)
After two confirmed U.S. cases of a superbug that thwarts a last-resort antibiotic, infectious disease experts say they expect more cases in coming months because the bacterial gene (mcr-1) behind it is likely far more widespread than previously believed...Army scientists...reported finding E. coli bacteria that harbor a gene which renders the antibiotic colistin useless...The mcr-1 superbug has been identified over the past six months in farm animals and people in about 20 countries...Health officials fear the mcr-1 gene, carried by a highly mobile piece of DNA called a plasmid, will soon be found in bacteria already resistant to all or virtually all other types of antibiotics, potentially making infections untreatable...Within the next two to three years, it's going to be fairly routine for infections to occur in the United States for which we have no (effective) drugs available...mcr-1 will find its way into carbapenem-resistant bacteria…the resulting virtually impervious bacterium would likely spread slowly inside the United States because CRE themselves are not yet widespread in the country, giving drugmakers some time to create new antibiotics...
- FDA defines the ability of compounders to make approved medicines (statnews.com)
In its latest (to) bid to set parameters around compounding, the US Food and Drug Administration...issued a pair of draft guidelines to clarify when compounding pharmacies are permitted to make versions of commercially available medicines...federal law currently states that a compounder generally shouldn’t make copies of drugs that are approved for sale, the agency is getting more specific and wants to ensure that a true clinical need exists before a compounded version of an approved medicine drug is made for a patient...A key issue has been the extent to which compounders can make medicines for patients in ways that may cross the line into more conventional drug manufacturing. The new law attempts to create boundaries, although the latest guidelines are designed to refine the FDA interpretation... the guidelines would permit compounders to make versions of brand-name drugs when a shortage exists or the brand-name medicine has been discontinued. Another example meeting the threshold of patient need might be a required change in formulation...But safety is not the only issue in play here. For instance, pointing to a lower price...a compounded version would be cheaper than a brand-name drug — is not justification for compounding an FDA-approved medicine...these are only draft guidelines — guidances in regulatory parlance — and are not set in stone.
- Advocates hope shaming drugmakers discourages price spikes (finance.yahoo.com)California Senate Bill 1010 (openstates.org)
Frustrated by the rising cost of prescription drugs, California health advocates hope sunlight and a dose of shame will discourage drugmakers from raising their prices too quickly or introducing new medications at prices that break the bank...They're promoting legislation that would require drugmakers to provide advance notice before making big price increases. Pharmaceutical companies have come out in force against the measure, warning it would lead to dangerous drug shortages...California's SB1010 would require pharmaceutical companies to provide advance notice to drug purchasers before increasing the price of a drug by 10 percent or $10,000 a year. For generics, the threshold is $100 a month or 25 percent. Insurance companies would be required to report data on drug prices to state regulators, including the portion of premiums attributable to pharmaceuticals...Proponents hope the advance notice will give governments, insurers and pharmacy benefit managers a chance to negotiate...But drugmakers warn it could create regional shortages of some drugs if large pharmacy chains or distributors horde medications to beat the price increase. That would create an environment for speculators to drive prices up, not down.
- More than 1 million OxyContin pills ended up in the hands of criminals and addicts. What the drugmaker knew (latimes.com)
In the waning days of summer in 2008, a convicted felon and his business partner leased office space on a seedy block near MacArthur Park. They set up a waiting room, hired an elderly physician and gave the place a name that sounded like an ordinary clinic: Lake Medical...The doctor began prescribing the opioid painkiller OxyContin – in extraordinary quantities. In a single week in September, she issued orders for 1,500 pills, more than entire pharmacies sold in a month. In October, it was 11,000 pills. By December, she had prescribed more than 73,000, with a street value of nearly $6 million...Purdue Pharma, the maker of OxyContin, tracked the surge in prescriptions. A sales manager went to check out the clinic and the company launched an investigation. It eventually concluded that Lake Medical was working with a corrupt pharmacy in Huntington Park to obtain large quantities of OxyContin.
- Sounding the alarm
- What Purdue knew
- Pills prescribed by Santiago
- Following the pills
- The skid row connection
- Pharmacist complaints
- Reports of concern
- ‘I was sitting on a gold mine.’
- ‘It really takes the ‘G’ a long time to catch up with these jokers’
- Drug maker loses battle over Canada’s right to impose a price cap (statnews.com)
A closely watched skirmish over the cost of prescription drugs has ended in defeat for a company that sought to challenge the right of a Canadian agency to impose a price cap on a pricey medicine...Canada’s Federal Court dismissed a constitutional challenge that Alexion Pharmaceuticals filed against the Patented Medicine Prices Review Board, which...sought to keep a lid on the cost of the company’s Soliris (eculizumab) medication...The...agency had asked Alexion to lower its price and repay sales generated by the drug from 2012 through the first half of 2014..."It is far from surprising," said Richard Gold, a professor at McGill University..."Alexion may still appeal in hopes that the Supreme Court of Canada will alter the law. I think the [possibility that the court will find] the provisions unconstitutional are remote. Parliament can create and limit patent rights as it wishes, but pharmaceutical companies have been known for being obstinate in the face of logic."