- U.S. court invalidates patent on J&J cancer drug Zytiga (reuters.com)
A U.S. administrative court...invalidated a Johnson & Johnson patent on its blockbuster prostate cancer drug Zytiga, bringing rivals closer to selling generic versions...The Patent Trial and Appeal Board said a Johnson & Johnson patent describing a method of administering Zytiga should not have been granted because the process it described was obvious...The ruling was a victory for U.S. generic drug company Argentum Pharmaceuticals LLC, which had asked the board to invalidate the patent in hopes of bringing its own version to market...The patent is due to expire in 2027. Argentum and other generic drug makers have been blocked from launching their own versions of the cancer drug until its expiration date...Zytiga generated nearly $2.3 billion in sales for Johnson & Johnson in 2016.
- Flu-pocalypse? Why scientists are scrambling to make a ‘universal influenza vaccine’ (foxbusiness.com)
Flu season is already shaping up to be one of the worst seasons in over a decade, claiming the lives of nearly 20 children across the U.S. and 7% of senior deaths over the last month, according to the Centers for Disease Control and Prevention...One of the reasons for the massive outbreak this year...is that it involves the dreaded H3N2, a strain of the influenza that isn’t “very well-matched” with the current vaccines that are being distributed across the U.S...In the making of the vaccine as it was being grown in eggs...it got mutilated a bit, so it drifted away from a really, really good match...even in a very good year the influenza vaccine is only about 60% effective. And, the projection of how effective it is going to be against the H3N2 this year is about 30%...Sanofi, who is one of the largest makers of flu vaccines...has been “continuously focused on improving influenza vaccines” over the years...This is evident in our introduction of Fluzone High-Dose Influenza vaccine, the first and only flu vaccine shown to have superior efficacy against influenza compared to Fluzone vaccine in adults 65 and older, and our recent acquisition of Protein Sciences, which has developed a recombinant DNA technology system for the production of recombinant proteins. The company additionally has ongoing efforts to develop a broadly protective influenza vaccine that would be effective despite natural mutation of flu strains over time...
- Universities accused of unethical ‘pick and mix’ reporting of drug trial results (telegraph.co.uk)
Universities have been accused of undertaking widespread unethical drug trials after The Daily Telegraph revealed Oxford researchers allowed a vaccine to be tested on babies despite doubts about its effectiveness...An analysis by the British Medical Journal cited a “systematic failure” to faithfully report the results of animal tests in order to secure funding and permission for human trials...The alleged practice potentially harms patients and risks stalling scientific progress by putting off investors...The investigation focuses on senior scientists at Oxford University who tested a candidate tuberculosis booster vaccine on monkeys at the Porton Down Government laboratory in Wiltshire in 2006 ahead of a trial involving nearly 2,800 South African babies...The animals inoculated with the drug, MVA85A, died at roughly the same rate as those exposed to TB without it, however this was not disclosed until after funding and permission for the wider infant clinical trial had been sought...Instead, the Oxford team reported that MVA85A had been successful in animal trials overall...Dr Deborah Cohen said the case highlights the “pick and mix” approach some researchers take to reporting the result of animal testing, which is far less stringently regulated than are human trials...
- Price competition to hit oncology next: BioPharma Dive’s 2018 predictions (biopharmadive.com)
Cancer drugs have largely remained unaffected by the still-simmering scrutiny over rising pharmaceutical prices, routinely costing over six figures for a year's treatment...drugmakers retain a free hand to debut new drugs at prices in line or above competitors, even without demonstrably superior efficacy...Yet that state of affairs could soon change. An influx of "me-too" drugs coupled with a lack of clear differentiation across several key classes could spur fitful price competition. That could open the door for payers looking to rein in costs, through more aggressive use of formularies...Lured by the promise of lucrative returns, biopharmas have invested heavily in oncology R&D, swelling pipelines with product candidates — many following in the footsteps of already approved drugs...For drugmakers, the risk is that assets like PD-1/L1 blockers or PARP inhibitors will become viewed as commodities. As more and more patients become eligible to receive these drugs, the crowding queue could trigger price competition in a way that oncology has so far sidestepped...
- Wal-Mart launches program to safely dispose of unused opioids (reuters.com)
Wal-Mart Stores Inc...will provide customers filling prescriptions for opioids with a packet of powder that will help them dispose of leftover medication in order to help curb misuse and abuse...The company said patients filling any new class II opioid prescriptions at its pharmacies will receive a free packet of the product - called DisposeRx - when filling a new prescription. Patients with chronic Class II opioid prescriptions will be offered a free packet every six months...In order to safely dispose of opioids, patients would add warm water and the DisposeRx powder to their pill bottle, which then forms a biodegradable gel around the pills.
- Big pharma is getting ready to spend tax reform dollars on big deals (investing.com)
There's a lot of anticipation for big-ticket pharmaceutical mergers and acquisitions to take off in 2018...That's in large part because of the tax reform that passed in 2017, which frees up cash companies have overseas and lowers the corporate tax rate..."We'd love to use the cash to buy and partner to expand our pipeline...There's a potential for another round of change within the pharmaceutical sector, given there's this much money overseas and that's got a chance to be repatriated...Cash waiting to be used...It's something that's on company's radars...especially as they think of ways to use that repatriated cash. Other options besides acquiring companies with new medications in the works include share repurchasing programs, increasing dividends, and in some cases paying down debt...But the changes that tax reform brings doesn't necessarily mean there's going to be an across-the-board flood of new deals, since some major pharmaceutical companies aren't based in the US to begin with.
- Judge deals setback to Cherokee Nation lawsuit over opioids (reuters.com)
A federal judge in Oklahoma has dealt a blow to a Cherokee Nation lawsuit seeking to stop the flow of addictive opioid painkillers in its territory by issuing a preliminary injunction to prevent the case from being heard in tribal court...District Judge Terence Kern ruled the tribal court lacked jurisdiction because the lawsuit involving six wholesale drug distributors and pharmacy operators does not directly concern tribal self-government...“While noting Defendants’ (Cherokee Nation‘s) evidence of the harm opioid abuse has caused to individual tribal members and families, and costs borne by the tribe, the Court cannot plausibly find that such harm is ‘catastrophic for tribal self-government’,” Kern said...The Cherokee Nation in April 2017 became the first major Native American tribe to seek redress in tribal court from wholesale drug distributors and pharmacy operators...The tribe said the highly addictive painkillers were saturating its territory and contributing to violence, delinquency and mortality...The companies said the lawsuit attempted to civilly enforce a federal statute, the Controlled Substances Act, under the guise of the tribe’s statutory and common law...
- FDA Delays Implementing Parts of ‘Intended Use’ Rule (raps.org)
Confusion and concerns over portions of a tobacco-related final rule have pushed the US Food and Drug Administration to delay implementation of the sections dealing with the types of evidence FDA may consider to determine how a manufacturer intends for its medical product to be used...This is a determination that can have significant implications for, among other things, how manufacturers communicate about and promote their products. How we determine intended use is an important issue that touches many fundamental aspects of the FDA’s work...FDA is seeking comments on the proposal to delay...effective date of the portion of the rule related to intended use of medical products, though the tobacco-related portions of the final rule will go into effect...At issue is how the initially proposed rule sought to remove a sentence, which FDA Law Blog last February called "the famous 'knowledge' sentence: 'But if a manufacturer knows, or has knowledge of facts that would give him notice, that a [drug or device] introduced into interstate commerce...is to be used for conditions, purposes, or uses other than the ones for which he offers it, he is required to provide adequate labeling for such a drug/device which accords with such other uses to which the article is to be put.'"...But the final rule did not remove the sentence and instead amended it with new language that angered industry groups, which called the move "a new and unsupported legal standard."
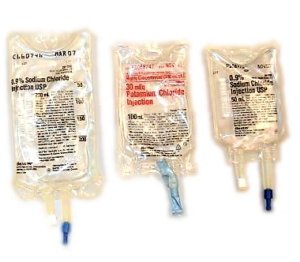
- ASHP Survey: IV Fluid Bag Shortage Affecting Patient Care (ptcommunity.com)
Pharmacists call shortage of small-volume parenteral solutions "severe"...The shortage...is affecting virtually all U.S. hospitals, according to results of a survey by the American Society of Health-System Pharmacists... that limited inventory of the critical product has required health care providers to adopt new procedures and use alternative therapies to treat patients...Survey respondents are taking a variety of steps to mitigate the shortages, including using alternative methods of administration such as intravenous push, intramuscular injections, or oral dosage forms; using nonformulary premixed solutions and/or frozen products; and implementing protocols that restrict the use of product...The shortage of SVPs is the latest in a series of shortages of critical medications, including sodium bicarbonate and epinephrine. Drug shortages pose a significant threat to the safety and quality of patient care in hospitals and other health care settings and may result in delayed treatment and increased risk of adverse reactions and medication errors...“We strongly believe that the current drug shortage situation is unacceptable and unsustainable,” said ASHP CEO Paul W. Abramowitz, PharmD...“It threatens harm to patients, wastes valuable health care resources, causes great uncertainty, and disrupts the health care system.”
- Replacing daily pills with a weekly regimen could help patients adhere to therapy (ptcommunity.com)
Replacing daily pills with a weekly regimen could help patients stick to their dosing schedule... Researchers at Massachusetts Institute of Technology and Brigham and Women’s Hospital have developed a capsule that can deliver a week’s worth of human immunodeficiency virus drugs in a single dose. This advance could make it much easier for patients to adhere to the strict schedule of dosing required for the drug cocktails used to fight the virus...The new capsule is designed so that patients can take it just once a week, and the drug will release gradually throughout the week. This type of delivery system could not only improve patients’ adherence to their treatment schedule but also be used by people at risk of HIV exposure to help prevent them from becoming infected...The MIT/BWH team is also working on adapting this technology to other diseases that could benefit from weekly drug dosing. Because of the way that the researchers designed the polymer arms of the capsule, it is fairly easy to swap different drugs in and out, they say. They are also working on capsules that could stay in the body for much longer periods of time...