- This Week in Managed Care: July 7, 2017 (ajmc.com)
Kelly Davio, Welcome to This Week in Managed Care from the Managed Markets News Network
- Walgreens, Rite Aid end $9.4 billion merger (pharmacist.com)
Walgreens Boots Alliance and Rite Aid have called off their $9.4 billion merger agreement. Instead, Walgreens says it will attempt to purchase one-half of Rite Aid's stores for $5.18 billion in cash. Executives said they crafted the smaller deal to address regulatory issues, but antitrust experts said it does not eliminate competition concerns. The Federal Trade Commission's review of the original Walgreens-Rite Aid transaction stretched 18 months, and the commission reportedly did not back away from concerns that the deal would have harmed competition. The commission worried about the merger's impact in regions where both companies have a strong store presence...Walgreens...said the smaller transaction addresses "all substantive" FTC concerns. The company will be adding stores in regions where it currently lacks a large presence, including the Northeast and MidAtlantic..."There's a chance that it won't go, that's the reality of the process. We believe it makes sense, we just have to wait until it plays out."
- Pharmacy Week in Review: June 30, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Lawyers Want the Testimony From OxyContin Company’s Ex-Chief (usnews.com)
A lawyer for one of the country's largest manufacturers of prescription painkillers says the public has "no right of access" to the testimony of its former president about how the company marketed the dangerously addictive OxyContin...The Kentucky attorney general's office sued Purdue Pharma in 2007, accusing it of misleading the public about just how addictive the opioid-based painkiller really was. They said that helped fuel a wave of addiction in Appalachia and beyond that cost the state millions of dollars in health care costs...Former Democratic Attorney General Jack Conway settled the case in 2015 for $24 million with an agreement to keep some court documents hidden from public view. They included a deposition from Richard Sackler, a former Purdue Pharma president and member of the family that still controls the company. His testimony could offer important insights into how much the company knew about the drug's addictive qualities.
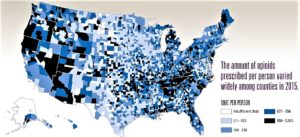
- Opioid prescribing is falling in the US, but not everywhere (philly.com)
Centers for Disease Control and Prevention...Prescriptions per capita, measured in morphine equivalents to account for various strengths and drug types, have declined steadily since their 2010 peak...Half the nation’s counties saw a decline (a quarter were stable) from 2010 to 2015. But there was wide variation, with providers in the highest-prescribing counties writing scripts for six times more opioids per resident than those in the lowest-prescribing counties...Nationally, the epidemic has been driven by high rates of addiction in more rural and white areas...Looking at county-level prescribing data helped CDC researchers to shed more light on some patterns of the painkiller epidemic. Counties with high rates tended to:
- Contain small cities or large towns (perhaps with pharmacies that drew residents from rural areas)
- Have higher percentages of white residents
- Have more dentists and primary-care physicians per capita
- Have more residents who were uninsured or unemployed
- Have more people with diabetes, arthritis, or a disability
But county-by-county analyses also illustrated how much is unknown about where and why opioids are given.
- Endo to pull opioid painkiller off U.S. market after FDA nudge (reuters.com)
Endo International Plc agreed...to remove its long-acting opioid painkiller from the U.S. market to comply with the health regulator's request last month, sending its shares down as much as 3 percent...The U.S. Food and Drug Administration in June had requested the withdrawal of Endo's Opana ER, marking the first time the agency asked for an opioid painkiller to be taken off the market for public health reasons...The regulator's request followed the recommendation of an independent panel of advisers, which concluded that the drug's benefits no longer outweighed its risks...
- New Pain Drug Likely To Face Price Challenges From Payers (forbes.com)
Centrexion Therapeutics announcing six-month data with its drug, CNTX-4975, for the treatment of moderate to severe knee osteoarthritis pain. The results are from a Phase 2b trial...and Centrexion is pretty excited by what has been seen so far:..The new data show large and statistically significant pain relief...a single 1 mg injection...resulted in large levels of pain reduction compared to baseline and statistical separation from placebo...These data represent the largest reductions seen in knee osteoarthritis reported for any drug treatment, marketed or in development…the active component is capsaicin...CNTX-4975 is "based on Centrexion’s proprietary STRATI technology (Synthetic TRans cApsaicin ultra-pure Injection), a highly potent, ultrapure, synthetic form of trans-capsaicin."...In certain ways, CNTX-4975 is reminiscent of the EpiPen. The active component of the latter is epinephrine, which, like capsaicin, is a drug that’s been known for decades. It’s the injector device, however, and not the drug, that drives Mylan’s proprietary position–and price–for the EpiPen. Similarly, the STRATI technology is what protects CNTX-4975 competitively, as anyone can easily access trans-capsaicin. In addition, epinephrine and trans-capsaicin are cheap drugs. These aren’t complex biologic molecules...The major cost will be for the STRATI technology...it is possible that Centrexion and its investors are going to demand premium pricing for the unprecedented pain relief...How will payers react?
- Shortages of Simple Drugs Thwart Treatments (wsj.com)ASHP - Drug Shortages List (ashp.org)
The scarcity of a simple medicine is complicating treatment for patients...Chronic drug shortages have burdened U.S. medical care for several years. Studies have estimated drug shortages add more than $400 million in annual U.S. health-care costs because hospitals and doctors are forced to seek more expensive alternatives, and health-care workers must spend extra time managing shortages...The shortages of low-cost medicines have been caused by a decline in the number of suppliers for certain products and failures by companies to build enough production capacity or meet manufacturing-quality standards...also...thinner profit margins for many older drugs reduce incentives for companies to invest in them...As a result of the shortage, doctors and hospitals are resorting to riskier and sometimes costlier treatments...FDA...said the agency works closely with manufacturers of drugs in short supply to help restore stocks, and asks other companies to increase production if they manufacture the same drug. The FDA says such efforts has helped cut the number of new shortages but can’t prevent all of them...
- This Week in Managed Care: June 30, 2017 (ajmc.com)
Kelly Davio, Welcome to This Week in Managed Care from the Managed Markets News Network
- Chinese courts call for death penalty for researchers who commit fraud (statnews.com)
An eye for an eye, a tooth for a tooth — a life for a lab book?...In the past few months, China has announced two new crackdowns on research misconduct — one of which could lead to executions for scientists who doctor their data...Scientists have been sounding alarms for years about the integrity of research in China. One recent survey estimated that 40 percent of biomedical papers by Chinese scholars were tainted by misconduct. Funding bodies there have in the past announced efforts to crack down on fraud, including clawing back money from scientists who cheat on their grants...Chinese...Ministry of Science and Technology proclaimed a "no tolerance" policy for research misconduct — although it’s not clear what that might look like...the mass retractions "seriously harmed the international reputation of our country’s scientific research and the dignity of Chinese scientists at large."...courts approved a new policy calling for stiff prison sentences for researchers who fabricate data in studies that lead to drug approvals. If the misconduct ends up harming people, then the punishment on the table even includes the death penalty...