- Computer-Simulated Tests Eyed at FDA to Cut Drug Approval Costs (bloomberg.com)
Computer simulations may get a role alongside human testing as part of an effort to bring new medications and medical devices to market more quickly and cheaply...The...Food and Drug Administration outlined a proposal...to help integrate computer modeling and virtual testing as part of the regulatory approval process for manufacturers -- a step the agency said could save money while helping find cures for puzzling conditions such as Alzheimer’s disease...The average cost of developing a new medication is about $2.56 billion...and much of that goes to fulfilling the FDA’s rigorous demands for proving safety and effectiveness...The price of new technology affects the ability of people to access these new treatments...We therefore need to be mindful of the costs of our regulatory processes, to the degree that these costs also affect the availability of new innovations, and the way that they are ultimately priced…The idea of computer simulated experiments, sometimes called in silico trials, has been around for years but the FDA hasn’t provided guidance to allow the drug industry to use it in testing...The FDA has begun using computer modeling to build databases to help researchers predict how new treatments...will perform. And the agency is developing a family of "virtual patients" for testing new devices…
- How AI Is Transforming Drug Creation (wsj.com)
Pharmaceutical companies hope computers can help them find new medications that are faster, cheaper—and more likely to be effective...The idea is that machines, which are adept at pattern recognition, can sift through vast amounts of new and existing genetic, metabolic and clinical information to unravel the complex biological networks that underpin diseases. That, in turn, can help identify medications likely to work in specific patient populations, while simultaneously steering companies away from drugs that are likely to fail...In the past, drug companies have used artificial intelligence to examine chemistry—whether a drug might bind to a particular protein, for instance. But now the trend is to use AI to probe biological systems to get clues about how a drug might affect a patient’s cells or tissues...Biological insights driven by machine learning also could help pharmaceutical companies better identify and recruit patients for clinical trials of therapies most likely to work for them, perhaps boosting the chances of those medications’ getting approved by regulatory agencies...The big difference between AI-driven drug trials and traditional ones...is "we’re not making any hypotheses up front. We’re not allowing [human] hypotheses to generate data. We’re using the patient-derived data to generate hypotheses."
- ICER to Work With VA on Drug Price Negotiations (raps.org)
In a first for the Institute for Clinical and Economic Review, which estimates the cost-effectiveness of drugs, the Department of Veterans Affairs Pharmacy Benefits Management Services office has agreed to use ICER drug assessment reports in drug coverage and price negotiations with the pharmaceutical industry...ICER says it will work with staff in the VA to integrate ICER reports into the VA formulary management process of evaluating the comparative clinical effectiveness and value of drugs...The VA requires that drug manufacturers offer a 24% discount off a drug’s average price or the lowest price paid by other (nonfederal) buyers, in addition to further discounts if a drug’s price outstrips inflation...The VA also directly negotiates lower prices with drug manufacturers, and with ICER’s help may establish a new norm for how the government can obtain lower-priced drugs...We will also assist the VA in exploring new ways it can use the reports to continue to provide a safe, effective and sustainable prescription benefit while more closely aligning a drug’s price with the value it provides to Veterans and the US taxpayer...ICER will also work with VA staff to develop options for using "value-based price benchmarks"...
- Pharmacy Week in Review: July 7, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Why Drugmakers Aren’t Sweating the Next Wave of Patent Losses (bloomberg.com)
Drugmakers plunged off a patent cliff earlier this decade, losing billions in sales as lucrative branded drugs lost exclusivity. An expensive lobbying effort aimed at preventing a repeat is paying off...The loss of a series of key patents for cholesterol fighters and other widely used medicines cost big-name drug companies about $82 billion in sales...forcing large-scale job cuts and a wave of deals to make up for lost revenue...Once again, the pharmaceutical industry is peering over the ledge. Over the next three years, roughly $60 billion of drug sales for companies including Roche Holding AG, Sanofi, and Eli Lilly & Co. are threatened by potential rivals…Roche may have the most at stake. It’s facing the loss of exclusivity for its three top-selling cancer drugs over the next two years, which account for more than $20 billion of its $51.4 billion in annual sales…Many of the drugs expected to lose their patent shields in coming years...are complex medicines produced by living cells, which makes replicating them more difficult...Drugmakers’ main lobbying groups, PhRMA and BIO, successfully watered down efforts to give biosimilars an easier approval process...Analysts say that given the longer approval arc for biosimilars, major drugmakers are less likely to be whipsawed by generic rivals when key medicines drop off patent. But over time, political pressure over high drug costs is expected to undercut the dominance of branded biologic drugs...
- Industry paid $8.2B to docs, hospitals last year (biopharmadive.com)
Drug and device makers collectively made nearly $8.2 billion in payments to physicians and teaching hospitals in the U.S. last year, according to data made public by the Centers for Medicare and Medicaid Services last week...Payments for research made up more than half of the total, which also includes money given to doctors and hospitals for expenses like travel, gifts, speaking fees and meals. The $8.2 billion recorded last year was a notch higher than 2015's total and $320 million more than 2014's figure...Among the ranks of big pharma...Roche spent the most — a princely $586 million in payments. Novartis and Pfizer were close behind with more than $475 million each, although both made more research-related payments than Roche did...The data gives a snapshot of the enormous sums of money paid by the drug and device industries to doctors and hospitals. Most of the money goes towards research, payments for things like enrolling patients into new clinical studies or study implementation. Research payments can also include direct compensation to doctors…CMS began tracking payments in 2013 as part of changes to federal law through the Affordable Care Act, with 2016 marking the third full year of the program...
- Change law and raise taxes to enable STPs to improve NHS, report urges (pharmaceutical-journal.com)
A lack of statutory power and severe underfunding are creating barriers to promising NHS reforms set out in sustainability and transformation plans (STPs), a think tank is warning...A report from the Institute for Public Policy Reform found that...44 regions in England had by and large correctly identified the right solutions, including moving care into the community and prevention...But there are several barriers that will prevent the ‘vision’ being realised, including a failure of leadership, the risk the NHS is being ‘engulfed by funding pressures’ and a lack of legal power to deliver the reform set out by the STPs...Among the IPPR recommendations is a call for the government to create a new ‘NHS tax’ by raising taxes for the highest paid to provide an extra £3.9bn a year to tackle the NHS ‘funding crisis’...In coming years, the NHS is facing a £22bn funding gap, which means limited resources are being spent maintaining the status quo rather than introducing a reform…There should also be changes to the law to better enable the pooling of budgets and commissioning functions locally and to give regional bodies a formal role in the system and put into effect place-based health and care...
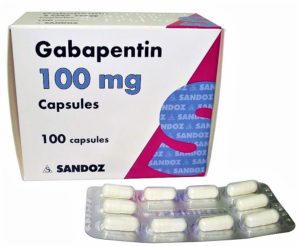
- New on the streets: Gabapentin, a drug for nerve pain, and a new target of abuse (statnews.com)
Ohio’s Board of Pharmacy began reporting sales of gabapentin prescriptions in its regular monitoring of controlled substances. The drug, which is not an opioid nor designated a controlled substance by federal authorities, is used to treat nerve pain. But the board found that it was the most prescribed medication on its list...surpassing oxycodone by more than 9 million doses. In February, the Ohio Substance Abuse Monitoring Network issued an alert regarding increasing misuse across the state...Gabapentin’s ability to tackle multiple ailments has helped make it one of the most popular medications in the U.S. In May, it was the fifth-most prescribed drug in the nation...law enforcement officials and drug counselors say the addition of gabapentin adds a new obstacle. As providers dole out the drug in mass quantities for conditions such as restless legs syndrome and alcoholism, it is being subverted to a drug of abuse. Gabapentin can enhance the euphoria caused by an opioid and stave off drug withdrawals. In addition, it can bypass the blocking effects of medications used for addiction treatment, enabling patients to get high while in recovery...
- Drugmakers and distributors face barrage of lawsuits over opioid epidemic (washingtonpost.com)
The companies that manufacture and distribute highly addictive painkillers are facing a barrage of lawsuits for the toll their product has taken on communities across the country as the worst drug epidemic in U.S. history continues to escalate...Within the past year, at least 25 states, cities and counties have filed civil cases against manufacturers, distributors and large drugstore chains that make up the $13 billion-a-year opioid industry. In the past few weeks alone, the attorneys general for Ohio and Missouri, along with the district attorneys for three counties in Tennessee, filed suits against the industry — and the attorney general for Oklahoma filed suit on Friday...The strategy echoes the effort against major tobacco companies in the 1990s and is born of similar frustration over rising death rates and the increasing costs of addressing the continuing public health crisis. After years of government and pharmaceutical firms failing to control the problem, some lawyers say the suits have the potential to force the industry to curb practices that contribute to it...Representatives of the companies deny wrongdoing and vow to vigorously defend themselves. They said they have taken steps to prevent the diversion of their drugs to the black market. Stemming the epidemic, they said, will take a coordinated effort by doctors, the industry, and federal and local government agencies..."As we look to prevent abuse and misuse in the future, it will require a forward-looking, systemic approach that calls on greater coordination and collaboration between health-care, law enforcement, and state and federal regulatory authorities," said the Healthcare Distribution Alliance, which represents companies that distribute drugs...
- Value-Based Care Increasing the Popularity of mHealth (ajpb.com)
Mobile health has become increasingly popular as the healthcare world moves towards value and away from volume, according to a new study by GBI Research. mHealth can connect patients with numerous healthcare services, while also collecting valuable data...This approach has already had an impact on the healthcare industry and numerous organizations are making investments in the technology. mHealth can also be used to encourage patients to adhere to their treatments, providing physicians with an additional way to communicate beneficial behaviors to their patients... advances in mobile technology, the expansion of communication, and the reduction in the cost of wireless technology...have huge potential to bridge the gap within existing healthcare systems, offering an alternative form of healthcare communication and treatment at a distance...The most important driver behind mHealth is the growing need for cost-effective ways to provide healthcare...Another important driver is the shift towards value-based care and reimbursement...healthcare providers are interested in helping patients achieve optimal outcomes rather than being incentivized to order more tests or perform more services.