- FDA moves to prevent Pharma from ‘gaming’ generic drug system (reuters.com)
The...Food and Drug Administration moved...to prevent pharmaceutical companies from "gaming" the system to block or delay entry of generic rivals…FDA Commissioner Scott Gottlieb said...that the agency plans to hold a public meeting...to identify ways pharmaceutical companies are using FDA rules to place obstacles in the way of generic competition..."We know that sometimes our regulatory rules might be 'gamed' in ways that may delay generic drug approvals beyond the time frame the law intended, in order to reduce competition,"..."We are actively looking at ways our rules are being used and, in some cases, misused."...These obstacles can include limiting the availability of branded products for testing by generic companies, or prolonging negotiations with generic companies over the implementation of shared risk-management programs...
- AMA calls for more transparent prescription costs (healio.com)
In response to the recent spike of many prescription drug prices, the AMA adopted several policies to provide patients with more information about the drugs that they are prescribed and a rationale for price increases…Taken together, these policies would bring much needed transparency to drug pricing and provide a clear benefit to consumers struggling with exorbitant costs…There seems to be no logic — or warning — to these price spikes…The AMA urged federal agencies to require that manufacturers list the suggested retail prices of medications on direct-to-consumer ads...In addition, AMA encouraged drug companies to notify the public of price increases of more than 10% for certain medications over a 12-month period...
- This Week in Managed Care: June 16, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Google bets on European biotech drugs, backs new fund (reuters.com)
Google is betting on the potential of European biotech companies to deliver life-changing drugs by investing alongside Swiss company Novartis in a new $300 million fund run by leading life sciences investment firm Medicxi...The move shows Google casting an increasingly wide net as it pumps cash into global medical research, seeding what it believes will become a core long-term healthcare business...The new fund will back both private and public firms with products that have already reached mid-stage Phase II clinical development, providing them with a new source of growth capital...There is a funding gap because there is a maturing class of biotechnology companies now in Europe...Europe boasts world-class universities and scientists, but its biotechnology sector has long been a poor relation to the bigger U.S. industry, where emerging life sciences firms are able to access a much deeper pool of capital...By providing funds for late-stage drug development, the hope is that more firms will be able to stay independent and continue to build up the value of their experimental medicines, rather than selling out prematurely to larger players...
- EU court: Vaccines can be blamed for illnesses without proof (ktvn.com)
The highest court of the European Union ruled…that courts can consider whether a vaccination led to someone developing an illness even when there is no scientific proof…The decision was issued…in relation to the case of a Frenchman known as Mr. J.W., who was immunized against hepatitis B in late 1998-99. About a year later, Mr. J.W. was diagnosed with multiple sclerosis. In 2006, he and his family sued vaccine-maker Sanofi Pasteur in an attempt to be compensated for the damage they claim he suffered due to the vaccine. Mr. J.W. died in 2011… the EU's top court said that despite the lack of scientific consensus on the issue, a vaccine could be considered defective if there was "specific and consistent evidence," including the time between a vaccine's administration and the onset of a disease, an individual's previous state of health, the lack of any family history of the disease and a significant number of reported cases of the disease occurring following vaccination.
- Drug Makers Being Probed by States Over Opioid Marketing (bloomberg.com)
A bipartisan group of state attorneys general announced...that they are jointly investigating the marketing and sales practices of drug companies that manufacture opioid painkillers at the center of a national addiction epidemic...Attorneys general from states including Massachusetts, Texas, Illinois and Pennsylvania announced the investigation two weeks after Ohio Attorney General Mike DeWine sued five drug manufacturers for misrepresenting the risks of opioids...We are looking into what role, if any, marketing and related practices might have played in the increasing prescription and use of these powerful and addictive drugs…Officials did not specify which companies were under investigation...
- Pharmacy Week in Review: June 16, 2017 (pharmacytimes.com)
Brian Bobby, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Will CVS Health’s Point-of-Sale Rebates Deflate the Gross-to-Net Bubble—and Disrupt the PBM Business? (drugchannels.net)
I examined the magnitude of the gross-to-net bubble—the growing spread between a manufacturer’s list price for a drug and the net price to a third-party payer after rebates...The bubble directly affects patients’ out-of-pocket costs. Many patients now have benefit plans with deductibles and are therefore required to pay the full, undiscounted cost of their prescriptions. Patients taking specialty drugs face large coinsurance computed using the undiscounted list price...One solution replaces formulary payments made to plan sponsors with prescription discounts for patients at the point of sale. Last week, CVS Health became the first pharmacy benefit manager to publicly embrace and explain this alternative benefit design...Do point-of-sale rebates solve the patients’ problems that the gross-to-net bubble causes? And will this benefit design mark the beginning of the end for the PBMs’ traditional economic model?
- FDA approves new, cheaper rival to EpiPen allergy shot (ktvn.com)
U.S. regulators have approved new competition for EpiPen, the emergency allergy medicine that made Mylan a poster child for pharmaceutical company greed...The Food and Drug Administration on Thursday approved Adamis Pharmaceuticals Corp.'s product, which should go on sale later this year...Symjepi is a syringe prefilled with the hormone epinephrine…Adamis says its product is easier to use than Mylan's EpiPen, a spring-loaded syringe filled with a set dose that comes with a training device...Symjepi also is smaller than EpiPen, so it's easier to fit in a pocket or purse. Most children and adults with severe food or insect allergies carry a device wherever they go and leave a spare at home, school or work...Just three years ago, EpiPens accounted for nearly 90 percent of both revenue and prescriptions filled in the U.S. for epinephrine injectors and syringes...In the first quarter of this year, brand-name EpiPens only drew about 60 percent of epinephrine device prescriptions, while generic EpiPens - mostly Mylan's - had captured 38 percent of prescriptions...
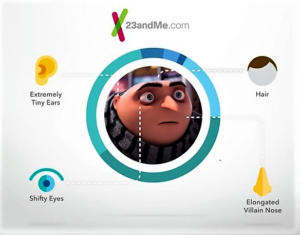
- 23andMe partners with ‘Despicable Me 3’ for first movie partnership (mmm-online.com)
The stars of the blockbuster "Despicable Me" movie franchise, the popular pill-shaped yellow Minions, have pitched everything from Amazon to Twinkies. This week, their evil overlord Gru is taking a starring role in an advertising campaign for 23andMe...Borrowing from the main plot point of the upcoming release of "Despicable Me 3," a 60-second commercial titled "Genetically Me" features the supervillain finding family through the results of his DNA analysis. In the film, Gru...discovers that he has a brother...through a stranger...23andMe teamed up with the $1 billion franchise for its first movie partnership in order to raise awareness about the brand and DNA testing...Personal genetics is a new and growing category. As a brand, we're tasked with not only building our brand and selling our product, but also building the category and explaining to people what it is, what they can get from it…