- Failure to Reauthorize User Fee Programs Would Result in About 3,000 FDA Layoffs (raps.org)
Representatives from the biotechnology, medical device and generic drug industries told members of the Senate Committee on Health, Education, Labor & Pensions...that if the five-year user fee programs are not reauthorized, the US Food and Drug Administration would likely see more than 3,000 job cuts...Those layoffs...would significantly curtail new medical product approvals for pharmaceutical, biotech and medical device companies and likely delay research...Senators on both sides of the aisle also criticized Trump’s budget proposal on Tuesday, which would cut billions from the National Institutes of Health and replace congressional appropriations at FDA with more user fees...Representatives from industry groups BIO, AdvaMed and AAM reiterated to the committee what FDA officials told the House Energy & Commerce Committee late last month: that a failure to act on the already-agreed-to reauthorizations would be devastating...
- 20,000 leagues for biotech to explore (biopharmadive.com)
The sea covers around 70% of the earth, and contains around 97% of the world's water. It's also home to almost 240,000 species...as a resource, it is still untapped...Bioprospecting is the discovery and development of new products based on resources from the natural world...We are not marine organisms, so until about 1970, no one even thought of the ocean. It was left as a deep secret. It seemed ridiculous to me that the ocean — with such a vast habitat — had escaped anyone's notice. But there are good reasons. People fear the ocean; it has been considered a very hostile, inhospitable place...the history of drugs from the sea isn't particularly long. Red algae have traditionally been used to make a treatment for colds, sore throats, chest infections including tuberculosis, kidney trouble, burns, cancer and indigestion...There are plenty of high-profile drugs that have gotten their origins from the sea…There are more than 25 marine-derived molecules in clinical trials, with over a thousand in pre-clinical development...There are still many societal challenges that the marine environment could help us to meet, such as antibiotic resistance...It is a source of chemical diversity, with novel targets and novel modes of action...
A snapshot of late stage development:
- PharmaMar...Plitidepsin and lurbinectedin, both derived from sea squirts... T-cell lymphoma...
- BeyondSpring...linabulin...is based on a marine fungus…non-small cell lung cancer...
- Ohr Pharmaceutical...Squalamine...found in dogfish shark tissues...a wide variety of indications…antimicrobial against bacteria, fungi, protozoa and viruses, as well as in cancer, macular degeneration, diabetic retinopathy and fibrodysplasia ossificans progressive...
- Wex Pharmaceuticals...puffer fish...neurotoxin tetrodotoxin...a non-addictive and rapid-acting pain killer without opioid-like side effects....
- 5 Takeaways From the AMCP Annual Meeting (ajmc.com)
At the Academy of Managed Care Pharmacy Annual Meeting, held March 27-30...in Denver...the cost of healthcare was on everyone’s minds and was a common thread in most sessions...Here are 5 key takeaways from the meeting.
- Value frameworks were on people’s minds...AMCP sees value frameworks as a tool that can help with the formulary decision making process…
- They are increasingly being used by payers...a survey of payers from the beginning of 2016 found that 26% were currently using value frameworks and an additional 22% planned to use them in the next 12 months. Only 19% had no plans to use value frameworks…
- Frameworks are just 1 solution centering on value…the National Pharmaceutical Council, outlined 3 other solutions that center around value. These include value-based insurance design, value-based contracting, and financing...
- Change is coming to healthcare...L.E.K. Consulting, followed the money in healthcare and came to the conclusion that healthcare is an unsustainably big business, which necessitates change.
- Obamacare repeal isn’t the only legislation to watch...Republicans may not have gotten the American Health Care Act, which would repeal and replace the Affordable Care Act, through the House of Representatives, but there could be future efforts to bring the legislation back in another form…
- Trump FDA Nominee Wants Lower Drug Costs With More Generics (bloomberg.com)Trump selects Scott Gottlieb as FDA nominee (cnn.com)
President Donald Trump’s pick to head the U.S. Food and Drug Administration is among the most vigorous advocates of lowering drug costs by approving cheap generics faster, an initiative aimed directly at the profit centers of major companies...Scott Gottlieb, a former FDA deputy commissioner, would make streamlining approvals his top priority...He’s particularly focused on complex medications that combine old drugs with newer delivery devices, as well as those with unusually complicated formulations...The administration could make the changes without Congress passing a law...But it also risks angering companies that have considerable legal firepower to defend their money-makers...Getting more generics to market faster could save Americans billions of dollars a year...Absent a new law, the FDA could do much to speed approval of generics. It could devise broad guidelines for copies. And the agency could become swifter in approving drug-device combos, finding ways to allow slight differences so long as they wouldn’t confuse patients...
- President’s Pledge
- Complicated Cures
- Pricey Pens
- EpiPen Dispute
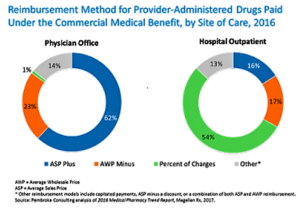
- Latest Data Show That Hospitals Are Still Specialty Drug Profiteers (drugchannels.net)
The new 2016 Medical Pharmacy Trend Report??...released...As always, the report includes a boatload of useful data on medical benefit spending and claims….Unfortunately, the latest edition also shows how hospitals continue to earn outrageous profits on specialty drugs. Commercial payers still use reimbursement approaches that permit hospitals to inflate specialty drug costs by thousands of dollars per claim...These hospital mark-ups translate directly into higher drug spending—regardless of how manufacturers set list prices. Using some fairly conservative assumptions about Remicade, I show below how hospitals are earning thousands of dollars more than the drug’s manufacturer does...Meanwhile, health plans are struggling to shift patients from hospital outpatient settings to physician offices—where costs are lower, at least until those sites are acquired by hospitals.
- Greece’s corruption prosecutor quits, citing pressure over Novartis bribery probe (fiercepharma.com)
Novartis is embroiled in a soap opera in Greece, complete with bribery allegations and a suicide threat. Now the plot is thickening. The country’s chief corruption prosecutor, Eleni Raikou, has resigned—and she’s blaming the Swiss pharma giant’s legal issues for her decision...Raikou...sending a letter to Greece’s Supreme Court claiming she was targeted by "unofficial power centres" over her investigation of Novartis...Greek authorities raided Novartis’ offices...after one of the company’s local managers reportedly made a suicide threat...The executive was one of the employees the authorities were interviewing...The probe began after media reports appeared alleging that Novartis had paid bribes to local "functionaries,"…Raikou’s resignation letter said that the bribery probe turned up "substantial and crucial evidence" that doctors and some politicians in Greece had received €28 million ($30.4 million) worth of bribes from a Novartis bank account...some of the evidence was linked to the prosecution of a local businessman and former minister of defense Yiannos Papantoniou...
- This Week in Managed Care: March 31, 2017 (ajmc.com)
Brielle Urciuoli, Welcome to This Week in Managed Care from the Managed Markets News Network.
- Sanofi Pasteur coughs up $19.8M to settle claims it overbilled the VA (fiercepharma.com)
Sanofi Pasteur agreed to pay more than $19.8 million to settle allegations that it overcharged the Department of Veterans Affairs for its products...The French pharma’s vaccines unit voluntarily reported the "calculation and reporting error" with the VA in 2012, and has since "cooperated fully and negotiated in good faith with the government,"...Sanofi Pasteur based its first disclosure to the U.S. government on products sold to the VA from 2007 to 2011. A follow-up investigation by the VA's Office of Inspector General found the overcharging error dated back to 2002, the DOJ said…This is not the first settlement in a pharma overpricing case. A range of drugmakers have paid settlements to wrap up improper billing allegations, which often fall under the False Claims Act, which governs overcharging for Medicare, Medicaid and other government programs. The most prominent recent case was Mylan’s $465 million deal to settle allegations that it overcharged Medicaid for EpiPens...
- Arizona Enacts Law: Pharmaceutical Companies to Legally Communicate Off-Label Treatment to Medical Professionals (policymed.com)
...pharmaceutical companies are not permitted to discuss off-label, legal, alternative uses for an approved drug with physicians and other healthcare professionals as regulated by the FDA. However, a bill in Arizona, HB 2382 that passed unanimously (The Free Speech in Medicine Act) has changed that...Governor Doug Ducey signed the bill into law, lifting the aforementioned off-label promotion restriction and allowing drug companies to communicate with doctors and other healthcare providers about safe and effective alternative uses for approved prescription drugs...The law...safeguards the free speech rights of pharmaceutical industry members to share truthful research and information about FDA-approved medicines...HB 2382...only protects the sharing of information that is "not misleading, not contrary to fact, and consistent with generally accepted scientific principles;" and it only applies to communication between pharmaceutical manufacturers and licensed healthcare professionals. The bill does not permit pharmaceutical manufacturers to advertise off-label uses directly to the public...Since Arizona enacted this protection for industry, it is likely to expand options in doctors’ toolkits, enhance patient autonomy, and increase access to healthcare. It will be interesting to see if any other states will follow suit and implement similar laws...
- Pharmacy Week in Review: March 31, 2017 (pharmacytimes.com)
Brielle Urciuoli, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.