- A Health-Monitoring Sticker Powered by Your Cell Phone (technologyreview.com)Battery-free, stretchable optoelectronic systems for wireless optical characterization of the skin (advances.sciencemag.org)
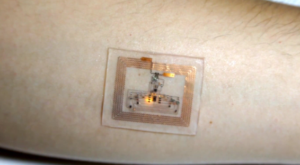
A wearable sensor measures blood oxygen using flashing LEDs. It's powered by radio waves from a nearby phone or tablet.
Stretchy electronics offer wearable health gadgets without batteries...Picture a health-monitoring patch you wear like a tattoo and that doesn’t need a battery...That’s the idea behind a demonstration by John Rogers, a stretchable electronics pioneer at the University of Illinois...whose lab created a stretchy skin patch that uses light pulses to monitor heart rate or sun exposure...it’s powered by a cell phone...with a near-field communications chip, the kind that’s used in apps like Apple Pay or for sharing photos between phones. That is, radio signals from a phone actually power the device and let it transmit information...Rogers says it means health-monitoring gadgets could be cheaper, smaller, and more lightweight than ever before...new device also measures heart rate and blood oxygenation using four LEDs to shine different colors of light into the skin. Changes to the color of the reflected light is picked up by photodetectors. A person’s heart rate is displayed as a flashing light.
- Doctor charged with insider trading while heading a clinical trial (statnews.com)
As a rule, principal investigators for clinical trials should not be trading in the stocks of companies whose drugs they are testing. But the feds allege that Dr. Edward Kosinski ignored this dictum and now faces charges of insider trading...Kosinski sold his shares in Regado Biosciences after receiving bad news about a clinical trial in which he was the principal investigator, according to...Securities and Exchange Commission. In the first stock trade, he avoided $160,000 in losses, and in the second transaction he made more than $3,000 by exercising options...He faces a maximum term of imprisonment of 20 years...alleged episode of insider trading is only the latest instance involving the pharmaceutical industry or those working with drug makers. The issue has increasingly raised concerns in connection with clinical trial work, as well as deal-making and the drug approval process, which some fear can be distorted by such activities.
- Pfizer Acquires Gene Therapy Firm Bamboo for $150 Million (bloomberg.com)
Pfizer Inc. said...that it acquired Bamboo Therapeutics Inc., a biotechnology firm developing gene therapies, for $150 million and potential milestone payments in a deal to expand the drug giant’s presence in the experimental field…Bamboo is developing treatments for neuromuscular conditions and central nervous system disorders. Pfizer...will take over the company’s treatments in development, which haven’t yet been tested in people, as well as Bamboo’s manufacturing facility. Bamboo’s most advanced drug, for a rare nerve-cell disorder called giant axonal neuropathy, is enrolling patients for its first human trial. It is also working on a treatment for Duchenne muscular dystrophy, a rare and deadly muscle disease.
- FDA warns Chinese drug maker over lying (statnews.com)
The next time that employees at Xiamen Origin Biotech want to lie to regulators about what they are doing, they may want to make sure that the doors to nearby rooms are closed...During an inspection last January of its facilities in the southeastern Chinese province of Fujian, an employee told a US Food and Drug Administration inspector that the company did not keep any drugs on location. But while they reviewed company operations in a conference room, the inspector happened to notice that an adjacent room was being used to warehouse relabeled medicines...The same Xiamen employee also thought nothing of telling the inspector that the company had stopped relabeling drugs in January 2015. But during the inspection, the FDA staffer reviewed a list of exported drugs that showed Xiamen had distributed them until January 2016...Pharmaceutical and ingredients manufacturers in China and India have come under intense scrutiny in recent years due to a series of disturbing events...the flow of FDA warning letters about companies based in these countries receives added attention, sometimes deservedly so. Xiamen, for instance, also lied to its customers, according to the FDA...The company falsified and omitted information on certificates of analysis...which are supposed to verify the veracity of its products...
- Business groups sue U.S. gov over tax rules that spoiled ‘Pfizergan’ (fiercepharma.com)
Pfizer and Allergan weren’t the only ones that were unhappy with the U.S. Treasury’s April move to block their $160 billion proposed megamerger. The U.S. Chamber of Commerce and one Texas business group were, too--and now they’re doing something about it...The pair sued the federal government...claiming that the Treasury Department’s move to tighten the reins on inversion deals violated the law...According to the allegations, the government tweaked the Internal Revenue Code itself after Congress refused to get on board with President Barack Obama’s inversion-limiting legislative changes--and the way Thomas Donohue, the chamber’s president and CEO, sees it, "this is not the way government is supposed to work."..."Instead of breaking the rules to punish companies engaged in lawful transactions, Washington should just do its job and comprehensively reform the tax code...The inversion crackdown that scuttled the Pfizergan transaction wasn’t the first of its kind. The April actions represented the third time the U.S. Treasury took matters into its own hands, with the first set--in October of 2014--toppling a proposed AbbVie-Shire merger....
- A key trial for a Bristol-Myers cancer drug fails in a ‘major surprise’ (statnews.com)
In a...setback for Bristol-Myers Squibb, its blockbuster Opdivo medicine failed to meet the primary endpoint in an important, late-stage study involving patients with newly diagnosed non-small cell lung cancer. The news sent Bristol-Myers stock plunging 17 percent...the top-line results from the Phase 3 trial...indicated that Opdivo failed to show progression-free survival in patients. By comparison, Merck disclosed two months ago that its own Keytruda treatment met the primary endpoint in a trial in which the drug was being tested for the same condition...Bristol-Myers has, so far, achieved...success with its immunotherapy treatments in combating various forms of cancer. The trial results are likely to undermine assumptions that the drug maker will maintain its leading role in treating lung cancer, the deadliest form of cancer...
- Pharmacy Business Transition Strategies – Being Prepared (pharmacypodcast.com)
Jimmy Neil talks with Dave Ellis VP – Pharmacy Transition Services at Cardinal Health...Pharmacy Transition Services is a full spectrum, consultative approach to the revolving world of pharmacy ownership for both buyers and sellers. We collaborate with pharmacy owners to develop a complete and actionable strategy, helping them to develop an exit plan that meets their financial goals, while preserving their legacy within the community...Assistance is also provided to those interested in acquiring pharmacies, but who may not understand the capital requirements or other requirements to achieve their goals. (podcast 25:54 min)
- Cephalon, U.S. states reach $125 million settlement over generic drugs (reuters.com)
Cephalon has reached a $125 million settlement with 48 states in connection with its alleged efforts to delay generic versions of its blockbuster sleep disorder drug Provigil from entering the market, New York Attorney General Eric Schneiderman said...The settlement...comes a little more than a year after the company struck a $1.2 billion parallel settlement with the Federal Trade Commission...The FTC accused the company of protecting its monopoly on Provigil by paying generic drug makers to drop their challenges to Cephalon's patent, in what is known as a "pay-for-delay" deal...the company successfully delayed generic competition of the drug by six years through filing patent infringement lawsuits and then settling them by paying competitions to delay selling generic versions of Provigil, Schneiderman said...When pharmaceutical companies put profits ahead of people by illegally restricting competition, it harms patients...
- Pharmacy groups advocate USP standards for biologics (chaindrugreview.com)
A coalition of pharmacy industry organizations has asked congressional leaders to weigh proposals that would free certain biologic drugs from adherence to U.S. Pharmacopeia standards...the American Pharmacists Association and eight other groups sent a letter to the chairman and ranking members of key House and Senate committees expressing concern about proposals by the Food and Drug Administration and a provision in Section 11 of the FDA and NIH Workforce Authorities Modernization Act that would exempt certain biologics — including biosimilars — from USP public standards for quality, including the naming of biologic and biosimilar medicines...USP’s work provides a uniform quality standard, clarity for health care practitioners and confidence for patients...Pharmacists rely on USP quality standards. Removing requirements to adhere to these standards will raise additional questions and concerns about the safety of these emerging products...Our organizations share a commitment to patient safety and, as such, we believe that biologics and biosimilars should be required to have the same nonproprietary names based on existing standards and mechanisms...the pharmacy industry groups said in the letter. "Given the broad and negative implications of such potential naming proposals, as well as their controversial nature, we are requesting that FDA not finalize current guidance and proposals and that legislation not include these provisions."
- This Week in Managed Care: August 6, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care, From the Managed Markets News Network.









