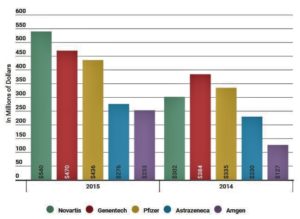
- These companies paid the most to docs last year (medcitynews.com)
The Physician Payment Sunshine Act was meant to shed light on the financial relationship between drugmakers, biotech, and medical device companies whether those payments be for general or research purposes...The Centers for Medicare and Medicaid Services has recorded these payments now for two complete years in a row with partial data available for 2013...The infographic below, created by MedCity News, based on data from CMS’s Open Payments website...shows the companies that spent the most money in 2015 in comparison with the amount they spent the year before and overall payments.
- Compounding Pharmacists Imprisoned for Dispensing Adulterated Drugs (pharmacytimes.com)Two Pharmacists Sentenced to Prison for Adulteration of Drugs in Connection with Alabama-Based Compounding Pharmacy (justice.gov)
Two compounding pharmacists from Alabama will spend a year in prison for distributing tainted drugs... The adulterated drugs were compounded at Advanced Specialty Pharmacy, which did business as Meds IV... David Allen...and William Timothy Rogers...pleaded guilty...to 2 misdemeanor violations of the Federal Food, Drug and Cosmetic Act...the 2...were sentenced to 12 months and 10 months in prison, respectively...also...1 year of supervised release after they get out of prison, and they’ll each have to pay a $5000 fine...Meds IV compounded...its own amino acid solution, then mixed it with other ingredients to create TPN (Total Parenteral Nutrition)...The amino acid solution happened to be contaminated with Serratia marcescens, and the TPN was prepared, packed, or held in insanitary conditions...the amino acid was prepared by Meds IV outside a laminar airflow workbench and was kept unrefrigerated, in a room that was not sterile, in a large pot sitting on the floor, sometimes overnight, before it was sterilized and used...Nine patients developed bloodstream infections and died, while others developed S. marcescens bloodstream infections but survived.
- Pfizer building modular biologics plant in China (fiercepharma.com)
Pfizer will build a biologics plant in China, where it will make biosimilars for the Chinese market but also for sale throughout the world. The New York drugmaker has turned to GE for construction of the facility, which has developed a modular construction process that will cut the cost and allow the plant to be operational in about 18 months, instead of three years...The $350 million facility, which it is building at the Hangzhou Economic Development Area in China, will be Pfizer’s third biologics production facility and its first in Asia. In addition to manufacturing, the facility will house Pfizer China’s Biosimilars and Biologics Quality, Technical Service, Logistics and Engineering divisions, and serve as a site for process development and clinical supplies. It will have about 150 employees when it is complete in 2018…The new center will be built using GE’s single-use technology…likened assembly to working with LEGO blocks...
- Public Citizen demands ‘black box’ warnings on gambling, sex urges for dopamine meds (fiercepharma.com)Reports of Pathological Gambling, Hypersexuality, and Compulsive Shopping Associated With Dopamine Receptor Agonist Drugs - abstract (archinte.jamanetwork.com)
A team of JAMA authors urged the FDA in 2014 to add "black box" warnings to dopamine agonists, linking the Parkinson's meds to pathological gambling, compulsive shopping and sexual obsessions...Public Citizen is upping the ante with a citizen petition to the agency, citing more than 80 studies supporting its claims...Public Citizen’s petition calls for stronger warnings on six FDA-approved medicines including GlaxoSmithKline’s Requip, Boehringer Ingelheim’s Mirapex and UCB’s Neupro. Used to treat Parkinson’s disease and restless leg syndrome, the medicines can cause uncontrollable urges that in turn lead patients to divorce, financial ruin and suicide attempts…
- This Week in Managed Care: July 2, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care, from the Managed Markets News Network.
- FDA warnings slam Chinese drugmakers, including ViiV partner (fiercepharma.com)
...two Chinese companies with ties to Western drugmakers have been called on the mat to account for problems in their manufacturing. The FDA has issued warning letters to plants operated by Shanghai Desano Chemical Pharmaceutical and Chongqing Lummy Pharmaceutical, slamming them both for manipulating testing and turning in falsified batch test results on APIs...
- Shanghai Desano Chemical Pharmaceutical - FDA...criticized the facility for conducting "unofficial" tests of drug batches that it kept out of its official record...also disturbed by finding many electronic logs of production deviations in a folder titled "GMP Anomalies" that had never been investigated…
- Chongqing Lummy Pharmaceutical...FDA...warning letter slams the drugmaker for widespread and serious data manipulation of batch analyses...In one egregious case, the FDA...an analyst set the gas chromatography personal computer clock back to make it appear as if testing had been done...7 months earlier. The analyst then performed 5 injections to produce falsified results for long-term stability for a finished API lot, deleted four and reported only the results of the final injection as passing in the quality-control...
- Magnetic blood clot dissolver could be 4,000 times more efficient than enzyme treatment (fiercepharma.com)
Researchers at ITMO University in St. Petersburg, Russia, have developed a magnetically controlled treatment designed to dissolve blood clots. The method looks to be a promising solution to some of the complications associated with enzyme-based thrombolytic drugs...To make the targeted drug, the scientists combined the mineral magnetite with the enzyme urokinase, commonly used as a thrombolytic agent. The nanosized particles can then be localized around a blood clot using an external magnetic field...The combination demonstrated up to 4,000 times more efficiency than the enzyme-based drugs alone...Now we are using a sledgehammer to crack a nut… In order to change the situation, we decided to develop a method of targeted drug delivery that would allow us to considerably reduce the dosage and ensure that the whole therapeutic effect is focused on the clot...
- Anthem, Express Scripts Face Legal Challenge Over Prescription Drug Prices (realclearhealth.com)Anthem sues Express Scripts for $15 billion over drug pricing (modernhealthcare.com)
Anthem and its pharmacy manager Express Scripts overcharged patients with job-based insurance for prescription drugs, alleges a lawsuit that seeks class action status for what could be tens of thousands of Americans...the latest wrinkle in a battle that has already pitted the major national insurer and its pharmacy benefit manager against each other in dueling legal actions...The case alleges that insured workers paid too much because Express Scripts charged "above competitive pricing levels" and Anthem, in effect, allowed those higher prices...Those actions...violate the firms’ responsibilities under a 1974 federal benefits law called the Employee Retirement Income Security Act...This action seeks to recover losses suffered by the plaintiffs…who overpaid and continue to overpay for the portions of the costs of prescription drugs…they are responsible for paying as plan participants," says the lawsuit, filed as Burnett v. Express Scripts and Anthem.
- Pharmacy Week in Review: June 30, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- After Medivation, what’s next? Alexion, BioMarin, Incyte could be Big Pharma’s next M&A targets: Analysts (fiercepharma.com)
Megamergers could fall out of favor over the next year...small- to mid-sized deals up to $25 billion? Expect those aplenty given “the need many larger companies have to try and boost their growth prospects.”
Medivation’s prized prostate cancer pill Xtandi
Incyte, a $14.7 billion market-cap company that boasts myelofibrosis med Jakafi.
Epacadostat is entering Phase III combo studies...is being studied in a range of other tumor types
Alexion, a $27 billion company whose Soliris--the priciest med in the world--leads a portfolio of three approved rare-disease drugs.
BioMarin, the perennial subject of takeover buzz with 5 marketed meds and a host of pipeline prospects.