- DEA Releases 2016 National Heroin Threat Assessment Summary (dea.gov)Drug Enforcement Administration released the 2016 National Heroin Threat Assessment Summary – Updated...The report outlines the expanding public health crisis afflicting America due to the use and abuse of heroin and other opioid drugs. Some key facts: (dea.gov)
Drug Enforcement Administration released the 2016 National Heroin Threat Assessment Summary – Updated...The report outlines the expanding public health crisis afflicting America due to the use and abuse of heroin and other opioid drugs. Some key facts:
- The number of people reporting current heroin use nearly tripled between 2007 (161,000) and 2014 (435,000).
- Deaths due to synthetic opioids, such as fentanyl and its analogues, increased 79 percent from 2013 to 2014.
- Deaths involving heroin more than tripled between 2010 (3,036) and 2014 (10,574) – a rate faster than other illicit drugs.
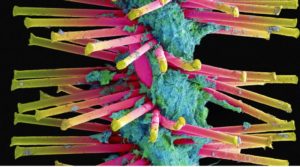
- Slimy clumps of bacteria kill thousands. Scientists are fighting back (statnews.com)
It’s a battle that seems ripped from a sci-fi film: Scientists are racing to develop new weapons to destroy the slimy colonies of bacteria, known as biofilms, that cause tens of thousands of deaths across the US each year...Biofilms are the leading cause of infections acquired in hospitals. They grow on medical devices such as heart valves, pacemakers, and catheters. They take root inside wounds, pulsing and rippling as they spread...Encased in gooey protective sheaths, biofilms are exceptionally hard to stop. Many are impervious to antibiotics. They also cost the health care system billions each year, as patients often require surgery to remove and replace contaminated implants...researchers and biotech startups are testing new methods of attack, from coating medical devices with spiky coverings to blasting bacteria with electrical fields to interrupting the chemicals that cells inside biofilm colonies use to send messages to each other...researchers at Ohio State announced they’d invented a way to coat the surfaces of medical devices with Y-shaped nanoparticles of quartz in a bid to block biofilms from latching on tight...
- Here’s a look at the battle:
- What are biofilms?
- Is this a new menace?
- Why are biofilms so hard to kill?
- So, what’s being done?
- Supreme Court rejects pharmacists’ religious claim (reuters.com)
A divided U.S. Supreme Court...rejected an appeal filed by pharmacists in Washington state who objected on religious grounds to providing emergency contraceptives to women...The justices, with three conservatives dissenting, left in place a July ruling by the San Francisco-based 9th U.S. Circuit Court of Appeals that upheld a state regulation that requires pharmacies to deliver all prescribed medicines in a timely manner...In Washington, the state permits a religiously objecting individual pharmacist to deny medicine, as long as another pharmacist working at the location provides timely delivery. The rules require a pharmacy to deliver all medicine, even if the owner objects...Alito said there is evidence the regulation was adopted because of "hostility to pharmacists whose religious beliefs regarding abortion and contraception are out of step with prevailing opinion in the state."..."If this is a sign of how religious liberty claims will be treated in the years ahead, those who value religious freedom have cause for great concern," Alito added.
- Drug industry overstates impact of patent reviews on innovation (statnews.com)
Drug makers complained bitterly...after the Supreme Court left intact a controversial procedure for reviewing patent disputes, arguing that the decision threatens valuable research efforts and that patients will eventually suffer. But the truth of those claims is debatable...The ruling upheld a process Congress created...for challenging patents outside the courts. It allows the Patent and Trademark Office to issue the "broadest reasonable interpretation" of patents. The case at hand had nothing to do with pharmaceuticals, but drug makers believe it will make their patents more easily challenged, and more likely to be overturned...The companies believe the procedure, known as inter partes review, is riskier than patent disputes decided in federal courts... Without the promise of effective patent rights, investments [in new medicines] would be far more difficult — if not impossible — to undertake...
- FDA approves a Gilead pill that is first to treat all forms of hepatitis C (statnews.com)
Gilead Sciences won regulatory approval...to sell a new hepatitis C combination drug, which can combat all six strains of the disease, and priced it below its older treatments...Epclusa, combines the older Sovaldi (sofosbuvir)medication with the newer velpatasvir, and costs $74,760 for a 12-week course of treatment, although that’s before any rebates are offered to payers. This is less than the list prices for both Sovaldi and Harvoni, another Gilead hepatitis C treatment...Gilead has argued its treatments are more affordable than paying for liver transplants, cancer, and hospitalizations. To what extent the newest drug will cause payers concern is unclear. Epclusa will cost roughly half of the most commonly used treatment regimen for patients with Genotype 3 of the disease...(which)...is...the most difficult to treat and one of the reasons Epclusa was granted priority review by the FDA...One consumer advocate charged the pricing for the new drug was out of line...Gilead still doesn’t get it — their pricing is outrageous. Once again, Gilead has shown that it is more concerned with protecting its profits rather than making its drugs available to all Americans...
- AstraZeneca sues FDA to prevent generic versions of Crestor (statnews.com)
Drug makers generally don’t complain when regulators widen the market for their medicines. But AstraZeneca filed a lawsuit...claiming the Food and Drug Administration is on the verge of illegally broadening the indication for its best-selling Crestor cholesterol pill, and the move would unfairly allow generic competition...The argument, which the company also made late last month in a citizen’s petition, hinges on the interpretation of federal law governing product labeling. Depending upon the outcome, AstraZeneca may either maintain a monopoly on Crestor for another seven years or face lower-cost rivals to a key revenue stream when the Crestor patent expires on July 8...the drug maker won FDA approval to sell Crestor to treat children with a rare genetic disorder called homozygous familial hypercholesterolemia...Under the Orphan Drug Act, the company was awarded an additional seven years of marketing exclusivity for Crestor, but only for treating this particular rare...disease...Several generic companies are lined up to sell a version of Crestor. AstraZeneca argues that a generic must include all pediatric labeling information approved for the corresponding brand-name drug. The company filed its lawsuit over concerns that the FDA will, instead, rely on a decision it made last year allowing generic companies to exclude certain information, so long as a safety risk is not created...AstraZeneca...believes federal law entitles the company to an additional exclusivity period of seven years for Crestor in the US...the American Journal of Clinical Oncology, a team of researchers argued that drug makers are exploiting loopholes in the Orphan Drug Act that allow them to widen the market for such drugs and distorting the original purpose of the law.
- Another big insurer will pay for key drugs based on patient outcomes (statnews.com)
Yet another health plan is aggressively pursuing deals with drug makers based on patient outcomes...Harvard Pilgrim Health Care...has reached agreements with Novartis and Eli Lilly to calibrate payments for two of their most important medicines...In one deal, the insurer will receive a discount from Novartis if its new Entresto (sacubitril/valsartan) treatment for congestive heart failure does not yield a specified drop in hospitalizations. In the other, Harvard Pilgrim agreed to accept a lower rebate from Lilly if its Trulicity (dulaglutide) diabetes drug lowers hemoglobin levels...Paying for value for drugs is an obvious way to go...The pharmaceutical industry is recognizing greater potential for regulation [on pricing], so I think they’re thinking more creatively and are more open-minded about how to develop contracts where cost is somehow related to outcome...The move toward so-called value-based contracting reflects accelerating costs for prescription drugs...It’s not rocket science to think about paying for value...Unfortunately, the pharmaceutical companies have been slow to adopt this...
- ‘Micro-hospitals’ offer alternative to urgent care model (fiercehealthcare.com)
Consumers like the convenience and accessibility of urgent care clinics. But hospitals may have found another model that better meets the needs of patients: the "micro-hospital."...While the state of Colorado prefers to call them "community hospitals," these licensed facilities offer emergency medical care, inpatient care, surgery, laboratory and radiology services...SCL Health...plans to open four locations in neighborhood settings with it's partner Emerus. The facilities are priced higher than urgent care centers, but less than a full-service hospital, and can treat a wider range of conditions because they have inpatient beds..."Micro-hospitals like this are more suited for large urban and suburban metro areas,"..."This model would probably be too large and complex for a rural market. It definitely is a trend...to create more accessible, cost-effective access points and alternative delivery models."...
- WV considers prescriber rankings in fight against opioid abuse (wvgazettemail.com)
A new way to curb the proliferation of prescription painkillers in West Virginia is in the works: "Prescriber report cards."...The state Board of Pharmacy is developing a system that will rank doctors by specialty based on the number of prescriptions they write for pain medications..."We’re going to categorize prescribers, and then send notifications of how they rank among their peers with their prescribing practices," said Michael Goff, a pharmacy board administrator. "It’s a way of telling them, ‘Hey, among other doctors in your field, doctors who do what you do, here’s where you rank.’"...One drawback: The report cards won’t be made public. State law requires such information to be kept confidential. Doctors would only see their own numerical ranking, not a complete list of rankings by specialty...
- UK pharma trade group on Brexit: ‘There’s a potentially negative story here’ (statnews.com)“UK must send strong signal it is open for business”, ABPI responds to Britain voting to leave the European Union (abpi.org.uk)
The aftermath from the Brexit vote in the United Kingdom is only first being digested the world over. As with so many other sectors, there are implications for the pharmaceutical industry. Some 70,000 people in the UK work for drug makers, including AstraZeneca and GlaxoSmithKline, which are based there. Will companies want to shift substantial portions of their operations elsewhere? The European Medicines Agency, which oversees product approvals, is also headquartered in London, raising questions about the ability of the UK’s own regulator, the Medicines and Healthcare products Regulatory Agency, to fill that role. Then there is concern about government support for basic research. We spoke with Michael Thompson, who heads the Association of the British Pharmaceutical Industry, the large trade group, about these unknowns...