- Novartis is accused of paying bribes in Turkey to boost prescriptions (statnews.com)Turkish prosecutor opens bribery probe into drugmaker Novartis (reuters.com)
An anonymous whistleblower has accused Novartis of paying bribes in Turkey by using a consulting firm to boost the use of its medicines...The alleged bribes reportedly resulted in $85 million in benefits to Novartis...The firm aided the drug maker by getting medicines added to formularies, or list of medicines that were approved for use in government-run hospitals...The consulting firm also helped Novartis avoid price cuts in other countries by winning government approval to change the names of two medicines...The Alp Aydin Consultancy was paid the equivalent of $290,000 plus costs in 2013 and 2014, but the relationship ended after the Turkish Social Security Institution began an investigation...A Novartis spokesman confirmed that an internal investigation is under way...We take any allegation of inappropriate behavior extremely seriously and investigate all allegations thoroughly...Over the past few years, a growing number of companies have been enmeshed in bribery scandals, some of which have resulted in large fines...Whether US authorities will investigate remains unclear...the allegations, which could not be independently confirmed, would also be passed along to the DOJ and the Securities and Exchange Commission...
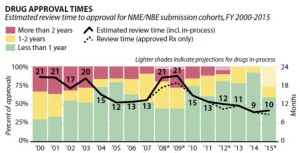
- FDA Review Times Steadily Decreasing, Report Finds (raps.org)
Despite wide variations across therapeutic areas, FDA review times for new drugs have steadily declined since 2009, according to a report released...by the California Life Sciences Association and Boston Consulting Group...Back in 2009, FDA was averaging 21 months for reviewing new molecular and biologic entities, but five years later, that average has been cut by more than half to nine months in 2014. In addition, the number of applicants waiting more than two years for an FDA review has fallen from more than 25% of applicants in 2000 to none by 2013.
- One year after Zarxio approval, future of biosimilars remains unclear (modernhealthcare.com)Biosimilar drugs could save up to $110 billion by 2020: IMS (reuters.com)
A year ago, providers, plans and pharmacy benefit managers thought they were on the brink of a new era of competitive drug prices. The federal approval of the first biosimilar for sale in the U.S. was supposed to foster new products that offered big discounts on some of the most expensive treatments...But there's been no flood of new drugs and no lower prices since the Food and Drug Administration's approval of Sandoz's drug Zarxio...an alternative to Amgen's cancer therapy Neupogen, remains the only biosimilar for sale in the U.S...Since January, 59 biosimilars...have enrolled in the FDA's Biosimilar Product Development Program...the agency was actively seeking to recruit additional staff to meet the demand...The agency has been struggling with how to address interchangeability, or the ability to switch a patient onto a biosimilar drug from an original biologic, and vice versa, without impacting safety or efficacy...Naming conventions is another hot-button issue...FDA...proposed adding a four-letter suffix to the non-proprietary names shared with brand-name biologics...All of these issues raise questions as to whether the biosimilar market could ever reach its potential...Dan Mendelson, president of Avalere Health, said the biosimilar market will eventually be worth tens of billions, but he expected growth to occur slowly in the U.S., as consumers get more comfortable with choosing such drugs over their originator counterparts...
- Substances Doubtful for Bulk Drug Substances List Could Be INDs (ashp.org)Individual Patient Expanded Access Applications: Form FDA 3926 (fda.gov)
Pharmacists, physicians, and advocacy groups that want patients to use substances unlikely to be on the upcoming "bulk drug substances list" for compounders should consider submitting "treatment" investigational new drug applications, FDA personnel recently suggested...FDA-cleared treatment IND applications...offer a legal workaround that can benefit many patients...a compounding pharmacy, could submit a treatment IND, which once that was in place could be expanded to treat a large number of patients...ASHP stated, absent "significant" revision, FDA's current expanded-access IND application process will not facilitate access to any drug available only from compounders...Jarow (Jonathan Jarow, from the Center for Drug Evaluation and Research)...acknowledged that submission of an IND application has seemed difficult to many individual healthcare providers seeking a drug for a single patient..."There's now a special form in development that has not been finalized—it's available in draft form—that caters to that specific type of IND rather than the general form that's used for all types of INDs, which looks very complicated," he said...Form FDA 3926, also known as "Individual Patient Expanded Access Investigational New Drug Application," went on display in February 2015 as part of a draft guidance for the pharmaceutical industry....
- Prominent doctors group supports controversial moves to lower drug prices (statnews.com)New Policy Paper Recommends Ways to Stem the Rising Cost of Prescription Drugs (acponline.org)
The rising cost of medicines has prompted yet another large group of physicians to speak out...The American College of Physicians, the largest medical society in the country, is calling for a set of familiar yet controversial actions to curb price hikes and improve patient access...For instance...believes that Americans should be allowed to import medicines from other countries...believes that drug makers should disclose their actual production costs, including research and development, used to set to pricing...Other ideas include allowing Medicare to negotiate prices with drug makers and expanding the mandate of the Patient-Centered Outcomes Research Institute...so that it can legally use a tool to evaluate the cost-effectiveness of medicines...The pharmaceutical industry regularly maintains that pricing funds needed investment and has previously argued against some of the proposals...Drug makers...maintain that importation would make it easier for counterfeit medicines to creep into the supply chain. Requiring companies to disclose R&D costs for each drug can be difficult to separate while such efforts overlook the role played by insurers and pharmacy benefit managers in setting prices...
- Jean Coutu launches Rx image transmission service (chaindrugreview.com)
Jean Coutu Group has introduced an image transmission service for new prescriptions that’s designed to make it easier for patients to fill medications...The Canadian drug chain said...that the new service is accessed via the Jean Coutu mobile application. To use the service, patients open their Health Record in the app and then take a photo of their prescription. The image is then sent directly to their Jean Coutu pharmacy. When the medication is ready for pickup, the pharmacy sends a text message to the patient...the image transmission service is a time-saver for both patients and their pharmacists. Prescriptions can be sent to the pharmacy as soon as patients get them from their doctors, and pharmacists can prepare the medications in advance...Besides the new prescription image transmission service, the Health Record also enables users to complete a simultaneous request for prescription renewals for one or more family members; receive an "it’s ready" notification via email or text when prescriptions are available for pickup...and scan a prescription label to request a refill.
- Pharmacy Benefit Management Tools Could Save Billions Over the Next Decade (ajpb.com)
Pharmacy benefit managers have been the target of scrutiny lately, in the media and even in proposed legislation. Detractors have called for more transparency for PBMs, accusing them of passing unnecessary drug costs along to plan sponsors and beneficiaries...To educate the public about the significant cost savings PBMs actually produce for plan sponsors and patients, the Pharmaceutical Care Management Association recently launched a national ad campaign..."This campaign is focused on educating policymakers and opinion leaders on how PBMs reduce costs, expand access and improve the quality of prescription drug..." said PCMA President and CEO Mark Merritt...today’s average level of PBM tool utilization yields a 10% to 20% cost savings for payers, and that a higher level of use could save payers an additional 10%.
- How Do PBM Tools Work?
- The Extent of PBM Tools in Current Practice
- Projected PBM Savings
- How Could PBM Savings Impact Employment?
- Pfizer, Allergan get request for additional information from FTC (reuters.com)
Pfizer Inc and Allergan Plc said they have received a request for additional information from the U.S. Federal Trade Commission with respect to their merger...The FTC's request for information extends the waiting period required under antitrust rules to 30 days, after the companies have provided the information...The companies said the request was anticipated as part of the regulatory process and they were working with the FTC in connection with the review.
- Shire gets favorable ruling against Allergan in Lialda patent case (reuters.com)
A U.S. district court...ruled in favor of Shire Plc, preventing Allergan Plc from selling generic versions of Lialda (mesalamine), the ulcerative colitis drug, in the United States until 2020...district court said that Allergan's Watson unit had infringed on two claims of the Lialda drug patent...
- Organ-on-a-chip startup Emulate raises $28.75M Series B (medcitynews.com)
Cambridge organ-on-a-chip startup Emulate has raised $28.75 million in a Series B round, with aims to commercialize its "Human Emulation System" on the R&D circuit. The idea, as with most lab-on-a-chip technologies, is to enhance in vitro research so as to make animal and in-human studies more efficient – using algorithms and microfluidics to accurately predict human response to medicines, chemicals and diseases...The financing will help expand Emulate’s organ-on-a-chip portfolio. Currently, it’s got organ emulations of the lung, liver, intestine and skin – but it plans to branch into organs like the kidney, heart and brain. Beyond that, however, is the concept of developing specific labs-on-chips to address important disease states, such as cancers, disorders of the intestine and microbiome, and infectious disease...Emulate wants its technology to operate as a plug-and-play system in the hands of product development teams at pharmaceutical, chemical, food and consumer products companies...