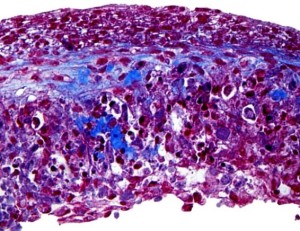
- Engineers 3D Print Tissue That Mimics How The Human Liver Functions (forbes.com)
Engineers at the University of California...say they have successfully 3D printed life-like liver tissue that simulates how the human liver functions and is structured. The researchers say the tissue could be used as a platform for drug screening...In the case of Federal Drug Administration approval for a drug, on average it takes around 11 to 14 years and $2.6 billion to get a drug to market...Around 90% of drugs don’t pass animal tests or human clinical trials. In the case of the new 3D printed tissue, the researchers say pharmaceutical companies could use the tissue as platform in the lab to focus on drugs that appear to be more promising and eliminate drugs that have less efficacy...To create the liver tissue that mimics real human liver tissue, the engineers created a diverse combination of liver cells and supporting cells systematically organized in a hexagonal pattern under a microscope. But to print that complex tissue, they needed a 3D printer that could accommodate the 3D micro-structures found in biological tissue. The team created their own bioprinting tech in the lab capable of reproducing the elements and features of the tissue…I think that this will serve as a great drug screening tool for pharmaceutical companies and that our 3D bioprinting technology opens the door for patient-specific organ printing in the future. The liver tissue constructed by this novel 3D printing technology will also be extremely useful in reproducing in vitro disease models such as hepatitis, cirrhosis and cancer...
- McKesson AccessHealth enhances suite of reimbursement tools (drugstorenews.com)
McKesson AccessHealth...expanded its suite of reimbursement and performance enhancement tools and relationships...While the shift to preferred networks and new reimbursement models is challenging for many independents, there are also opportunities for incentives...There is a tremendous chance for innovative pharmacies to leverage their strengths, improve their overall pharmacy performance and maximize their reimbursement by interacting with their patients and providing care...more than one-third of Medicare D claims through AccessHealth contracts will include performance-based reimbursement incentives...those pharmacies or pharmacy networks who display top performance will be charged a smaller...Direct and Indirect Remuneration, amount than those who underperform. This...presents independent pharmacies with an...opportunity to reduce their DIR charges, thereby maximizing their potential reimbursement. The new suite of tools include:
- AccessHealth Pharmacy Performance Guidebook...offers detailed instructions on how to develop action plans to improve patient behavior and boost outcomes using such tools as medication therapy management case completion, refill reminder programs, medication synchronization and behavioral coaching;
- AccessHealth DIR Estimator Tool...allows a pharmacy to enter information specific to them and estimate accrual and incentive amounts that may impact DIR payments in order to plan accordingly;
- AccessHealth Webinar Series...designed to help independent pharmacies prepare for new reimbursement rates as well as identify opportunities to leverage the movement toward pay-for-performance incentives.
- U.K. cost gatekeepers say ‘show me’ to Alexion’s new rare disease med Kanuma (fiercepharma.com)
The U.K.'s cost-effectiveness gatekeepers think Alexion's newest rare-disease drug, Kanuma (sebelipase alfa), may be worth using in babies with lysosomal acid lipase deficiency. But it's going to need to see more research on the pricey med's costs and benefits before it shells out...At £491,992 ($704,630), the therapy doesn't represent good value for all patients, the National Institute of Heath and Care Excellence said in draft guidance. But there's still time for the company to change the watchdog's mind before it makes a final decision...Alexion...said in a statement that NICE had "failed to recognize the transformative clinical innovation of Kanuma" as a treatment for LAL deficiency, which can be particularly serious in babies. The inherited genetic disease causes fat to build up in cells, with survival for babies pegged at less than 12 months...it'll be up to the...drugmaker to prove the treatment's worth...
- Community pharmacy reforms (rpharms.com)
Changes to community pharmacy in 2016/17 and beyond...In a letter to the Pharmaceutical Services Negotiating Committee...the Department of Health and NHS England announced there would be a...reduction in funding through the community pharmacy contractual framework...this reduction in funding will have a substantial impact on pharmacy business owners, their employees and locums...it was announced that funding...would be cut by at least 6%, equating to £170 million ($242 million); lowering the funding available through the community pharmacy contractual framework from £2.8 billion ($4 billion) to £2.63 billion (3.75 billion). It is anticipated these changes will take effect in October 2016...Alongside the cut in funding...further changes that the Government would like to see, including:
- To better integrate pharmacy into the wider primary care and community health system – such as closer working with GP surgeries, care homes and urgent and emergency care departments - so that patients can benefit from pharmacists’ clinical skills through a "Pharmacy Integration Fund".
- A "Pharmacy Access Fund", which would provide more NHS funds to certain pharmacies compared to others, considering factors such as location and the health needs of the local population.
- The potential for automation and centralising dispensing to provide efficiencies. Also, in some areas, the distribution and number of pharmacies does not match local patients' needs.
- The view of the Government that patients would like to order prescriptions online and that "click and collect" and delivery options would be easier to arrange.
- ObamaCare May Be Growing The Number Of Unpaid Medical Bills (forbes.com)
A big hospital chain’s surprise decision to write off a slug of bad debt may be a signal of much deeper consumer healthcare strains being caused by ObamaCare...Community Health Systems surprised analysts this week...the company would take a $169 million provision for bad debt. The write off was a big part of Community’s dismal fourth quarter earnings report...The rising amount of uncollected co-pays and deductibles may be an early sign of consumer stress as the economy weakens. But more likely, it also reflects changes in the healthcare market that are saddling consumers with a much bigger share of their medical costs...ObamaCare is playing a big role...The structure of the insurance products offered under ObamaCare was deliberately skewed toward hollowed-out health plans. These plans sport large out-of-pocket limits and often skimpy or no co-insurance on drugs and doctors purchased outside a health plan’s increasingly narrow drug formularies and provider networks...Americans are now less insured for medical costs…The earnings report from Community Health was an early financial harbinger of this trend. Collecting on these rising out of pocket costs will get more difficult for healthcare providers as the ObamaCare insurance designs become the new market standard. This will be ObamaCare’s legacy – a new standard for hollow health coverage.
- Pharmacy Week in Review: February 19, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Teva Said to Seek Fast EU Approval for Allergan Unit Bid (bloomberg.com)
Teva Pharmaceutical Industries Ltd. will try to win an early European Union approval for its takeover of Allergan Plc’s generics unit by making concessions to regulators this week...Teva and Allergan will meet a deadline to propose remedies aimed at allaying any EU antitrust concerns...If EU regulators are satisfied with the proposals, they could clear the transaction without an extended probe that can last about 90 working days. The companies plan to close the deal by the end of March...Teva’s $40.5 billion cash-and-stock bid for Allergan’s generics drugs business will make it the world’s largest maker of generic medicines, giving it greater negotiating power with governments and private-health insurers. Allergan will receive a $1 billion termination fee from Teva if the deal fails to close due to regulatory issues.
- Why You Should Watch Zubin Damania Innovate Healthcare (medcitynews.com)Blank Script | Taylor Swift (youtube.com)Big Pharma | Big Poppa Parody by Biggie | ZDoggMD.com (youtube.com)
You may not know the name of one of the most talented and innovative forces in healthcare but there is a high probability that you have seen one of his video parodies about health. This is because Zubin Damania, M.D., goes under the alter ego ZDogg MD...He is creator of ZDogg MD and the Founder and CEO of Turntable Health, a direct primary care clinic in Downtown Las Vegas...He has been called the Weird Al of healthcare and uses humor and pop culture to education populations about important health issues... So by creating this outlet for humor, satire, and creativity, I sort of found that unique part of my voice that had been suppressed, and I was able to feel like I was reaching people and effecting real change that I couldn’t do in the hospital...As a hospitalist in practice for 10 years, I was deep in the trenches of medicine, deep in the daily paradox of joy and horror...So I speak the language of that world, a language that emerges from the shared experience of healthcare people. Very often this tribe feels voiceless, so my goal has always been to find a new way of expressing not just our dissatisfaction, but our vision for how to make things better...Payment models and primary care are at the top of my list...Fix that base of the pyramid and other stuff will fall in place. Fixing that culture is crucial...
- Canadian provinces close the door on Alexion over price of rare disease drug (statnews.com)
A simmering battle in Canada over the cost of a rare disease drug took a new twist as a government entity ended talks with the manufacturer because it failed to agree on evidence for justifying the price...The pan-Canadian Pharmaceutical Alliance, which negotiates drug prices on behalf of provinces and territories, was holding coverage talks with Alexion Pharmaceuticals over its Soliris (eculizumab) treatment for six months before reaching a dead end. The medicine costs up to $525,000...Patients and physicians want to try anything that may work to help them cope with their disease...public drug plans cannot provide coverage for all individuals who may wish to try Soliris or any drug — regardless of its cost — where the clinical evidence does not demonstrate improved health outcomes...Soliris is used to combat paroxysmal nocturnal hemoglobinuria...a genetic disease that destroys red blood cells, and also atypical hemolytic uremic syndrome...which is a progressive and life-threatening disease affecting the immune system...Alexion...negotiated in good faith and had not received any feedback until the decision to end talks was made...the UK government watchdog agency for cost-effectiveness, which is notoriously stringent about assessing drug values, last year recommended coverage for Soliris...
- WHO-backed pharmacovigilance program in India lags (fiercepharmaasia.com)
India's program to track the side effects of drugs in patients around the country is being hindered by low staff, lack of equipment and poor funding...India's pharmacovigilance program...currently sends data that medical professional collect of drug side effects to VigiBase--the database of the World Health Organization--as part of a global initiative to improve drug safety...Low staff numbers and poor equipment, as well as few properly trained personnel, have hampered the collection of this data...These important details about drug reactions could be dangerous to miss...The lack of quantity and quality in drug reaction data means India's government has less information on judging whether certain drugs have adverse side effects. Though the number of reports has risen...India has a long way to go to get their pharmacovigilance up to speed...