- Drug Price Hikes and Shortages Have Similar Roots, Experts Say (ashp.org)
Experts say that recent increases in the prices of off-patent generic drugs are closely tied to the drug shortages that have plagued hospitals for years...They have the same root cause: a lack of competition in certain parts of the generic market...The first indication we had of problems in the generic market were not prices; they were shortages. Hospitals were having serious problems filling prescriptions...The second manifestation we're getting now is the higher prices for certain generic drugs...without competition, drug companies can raise their prices several thousand percent simply overnight...arbitrary and unpredictable inflation is not sustainable for our hospitals, especially when we receive capitated payments for most of our patients...hospitals handle such high prices in the same way they do drug shortages: by rationing drugs, stocking smaller quantities, and devoting "huge hours of manpower" to managing the problem...the business strategy driving the price hikes affects a relatively small number of drugs but is destabilizing reimbursement practices surrounding those medications...compounding pharmacies could play a greater role in preventing price hikes affecting off-patent drugs...Other potential solutions...included speeding FDA's review of marketing applications for older generic drugs that have no competition in the marketplace and instituting price regulations for these drugs akin to the regulations that affect public utilities...
- I.V. Batching Improvements Cited in Baldrige Win (ashp.org)
The health system that won a 2015 Malcolm Baldrige National Quality Award for healthcare credited improvements in i.v. medication preparation as a contributing factor in earning the recognition for performance excellence...We are constantly looking at new and better processes throughout all of our departments," said Brian Sayre, director of pharmacy for Charleston Area Medical Center Health System in West Virginia... the health system had greatly reduced its drug costs by using principles of lean methodology to overhaul the batching of i.v. medications...Lean methods focus on improving services and reducing waste and have been used in many types of business operations. Six Sigma, a system that complements lean processes, is a disciplined, data-driven strategy for eliminating defects in manufacturing and other processes..."Lean principles [and] Six Sigma principles fit very nicely into pharmacy workflow processes," said Sayre...the pharmacy staff calculated that switching from one daily cart fill to five cart fills per day would reduce i.v. drug waste..."The process...saved $134,000 annually, exceeding a target of a 50% reduction,"
- FDA faulted for failure to track safety issues with drugs already on market (statnews.com)
Most Americans assume that drugs approved by the Food and Drug Administration are safe to take as directed. But safety concerns often arise only after the drugs go on the market, when companies or doctors tell the FDA about cases of patients who have fallen ill or died from their medications...a federal watchdog agency said the FDA is failing to sufficiently track and publicly disclose instances in such cases...Government Accountability Office investigation...raises...concerns about the FDA’s oversight. It expresses particular concern about the lack of tracking of drugs cleared under two expedited approval programs, which account for about one-quarter of all medicines permitted to go on the market...investigators also criticized the FDA for failing to post quarterly reports listing certain potential safety issues that it has identified. Despite a statutory requirement that it do so, last year FDA posted no reports at all in its tracking system...FDA lacks fundamental resources and leadership in ensuring that drugs brought quickly to market are truly safe and effective,”...“If FDA is shifting more of the safety risk to consumers by allowing fewer and shorter clinical trials on expedited drugs, adequate tracking of drug safety issues and review of post market studies are absolutely vital.”...The backlog of postmarket data has been a recurring problem at the FDA...
- French drug trial disaster leaves one brain dead, five injured (reuters.com) News Release (bial.com)
One person has been left brain dead and five others have been hospitalized after taking part in a clinical trial in France of an experimental drug made by Portuguese drug company Bial, French Health Minister Marisol Touraine said...90 people have taken part in the trial, taking some dosage of the drug aimed at tackling mood and anxiety issues, as well as movement coordination disorders linked to neurological issues...The six men aged 28 to 49 had been in good health until taking the oral medication at the Biotrial private facility that specializes in clinical trials..."This is unprecedented"..."We'll do everything to understand what happened."...The brain-dead volunteer was admitted to hospital on Monday...For three of the five others...there are fears of irreversible handicap...One of the six had no symptoms but was being carefully monitored...The medicine involved is a so-called FAAH inhibitor that works by targeting the body's endocannabinoid system, which is also responsible for the human response to cannabis...Bial said in a statement it was committed to ensuring the wellbeing of test participants and was working with authorities to discover the cause of the injuries, adding that the clinical trial have been approved by French regulators.
- Major drugmakers push back in U.S. price debate (reuters.com)
With a backlash brewing over the price of medicines in the United States, drugmakers are pushing back with a new message: Most people don't pay retail...Top executives from Eli Lilly and Co, Merck & Co and Biogen Inc said in interviews...this week that the media focus on retail, or "list prices," for branded medications is misplaced...They stressed that the actual prices paid by prescription benefit managers, insurers and other large purchasers are reduced through negotiated discounts...But the industry practice of raising prices each year for treatments used by millions of people is attracting new attention...the industry needs to better explain the value of drugs and how they can prevent healthcare costs down the line...We have to explain the difference between the list price and the net price…Drugmakers keep actual pricing details close to guard their position in negotiations with commercial insurers and government health plans like Medicaid. There is no centralized catalog of U.S. list prices or rebates for medicines...U.S. health insurers say that, even accounting for discounts, drug prices are rising at an unsustainable rate, and they are pressuring drugmakers for cuts.
- 4 Pharma Market Trends to Watch in 2016 (pharmacytimes.com)
Several pharmaceutical market trends are likely to affect pharmacists’ practice this year...The Pharmacy Forecast 2016-2020 from the American Society of Health-System Pharmacists Foundation analyzed pharmacy trends and described strategies for health-system pharmacists to keep pace with the evolving scope of their practice...Pharmacists and health systems must recognize, plan for, and appropriately react to dynamic trends in the pharmaceutical marketplace to ensure that they are positioned to continue providing the best patient care...Here are 4 pharmaceutical market trends to watch in the year ahead:
- Generic Drug Pricing
- Enforced Product Tracing
- Specialty Drug Spending
- Limited Drug Distribution Channels
- Pharmacy Week in Review: January 15, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program highlights the latest in pharmacy news, product news, and more.
- New Recertification Deadlines for Active CPhTs (ptcb.org)
This is a reminder that effective in January 2016, PTCB has changed the recertification schedule for active PTCB Certified Pharmacy Technicians (CPhTs). All CPhTs are affected by the changes. Most active CPhTs have a new certification expiration date, while a small percentage are keeping the same expiration date. All are required to apply for recertification by the first day of their expiration month or they will be charged a late application processing fee of $25...New Window: The new ‘Recertification Window’ opens 60 days (formerly 100 days) before a CPhT’s certification expiration date...CPhTs may check their certification expiration dates by logging into their PTCB Account...
- Merck to pay $830M in Vioxx securities lawsuit (cnbc.com)
Merck...said it would pay $830 million to settle a federal class action lawsuit involving allegations the company failed to adequately inform investors about heart risks from its now-recalled Vioxx (rofecoxib) pain medication....The drug...became a blockbuster product...But the company in 2004 recalled Vioxx from the market after a colon-polyp prevention study showed it more than doubled the risk of heart attacks or stroke after 18 months of use...After thousands of former users of Vioxx sued Merck, claiming they had been harmed by the drug, Merck in 2008 settled most remaining product-liability lawsuits for $4.85 billion...other litigation continued, including a multi-district class action lawsuit...Merck kept investors in the dark about negative data from earlier company-sponsored trials of the medicine...The company's cash payment for the settlement and fees will be about $680 million...company said it still faces other individual securities lawsuits related to Vioxx.
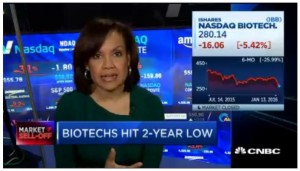
- Biotech leads Nasdaq lower (video.cnbc.com)Best names to buy after biotech sell-off: Pro (video.cnbc.com)Is it time to bet on biotech? (video.cnbc.com)
CNBC's Bertha Coombs highlights the big Nasdaq laggards as biotech concerns weigh.