- Synthetic drugs pose alarming U.S. overdose risk: DEA chief (reuters.com)
Synthetic designer drugs, especially synthetic opioids like fentanyl implicated in the death of pop star Prince, pose an "unprecedented" threat for U.S. overdoses and deaths, especially among youth...Drug Enforcement Administration has been cracking down on synthetic drugs since they began gaining popularity in 2010. But because each newly designed drug needs to be separately banned through a "clunky and cumbersome" process, the problem continues to spiral, DEA head Chuck Rosenberg told a U.S. Senate committee...For every one substance we’ve controlled, legislatively or administratively, there are 11 more out there that are uncontrolled...Rosenberg testified that the DEA has seen a particularly alarming rise in illicit use of the fentanyl...More than 11 million people illegally consume prescription painkillers for non-medical purposes each year, and overdoses are likely to increase as fentanyl is introduced to that market...
- Website Domains of Fake Online Drug Outlets Seized by Federal Authorities (nabp.net)Federal Court Orders Seizure of 67 Website Domains Involved in Smuggling and Selling Misbranded and Counterfeit Prescription Drugs (justice.gov)
Sixty-seven domain names of commercial websites involved in the illegal sale and distribution of counterfeit and prescription drugs had seizure orders executed against them, according to an announcement by the US Department of Justice. The federal government now has custody of the seized domains. When visiting the sites, users will see a seizure banner indicating that the domain name has been seized for violation of federal laws against smuggling and trademark misuse...Individuals who have an interest in the seized domain names have a period of time after the Notice of Seizure to file a petition with a federal court and additional time after the notice to contest the forfeiture.
- Drugmakers can’t charge beyond making costs for compassionate use: FDA (reuters.com)
Food and Drug Administration said companies could only charge patients for the cost of manufacturing experimental treatments used under compassionate grounds, and it cannot force government or private health insurers to pay for these drugs..."Compassionate use" of experimental drugs allows physicians to prescribe unapproved treatments for patients who have no other satisfactory alternatives in the market...The FDA's move seems to be intended to soften the repercussions of its possible rejection of Sarepta Therapeutics Inc's muscle-wasting drug (Duchenne muscular dystrophy)...FDA last week deferred its decision on whether to approve Sarepta's drug, eteplirsen, after an advisory panel determined that the treatment was not effective...The FDA is trying to create a compromise, saying drug companies can charge for a drug even if its not approved...Sarepta's drug has been in the spotlight over the past few months with patient groups and parents arguing passionately in favor of the treatment to pressure the regulator to approve the drug.
- India’s Sun Pharma gets U.S. subpoena over generic drugs pricing (finance.yahoo.com)
U.S. Department of Justice has subpoenaed India's largest drugmaker Sun Pharmaceutical Industries Ltd seeking information about the pricing and marketing of the generic drugs it sells in the United States...The DoJ's antitrust division has also asked Sun Pharma's U.S. unit for documents related to employee and corporate records and communications with competitors...They specifically cited doxycycline hyclate 100 milligram, an antibiotic for which the price doubled in the year through June 2014...Other generic drugmakers including India's Dr Reddy's Laboratories Ltd and U.S. firm Allergan Plc also received subpoenas from regulators seeking similar information last year, but they did not disclose the names of the products involved...
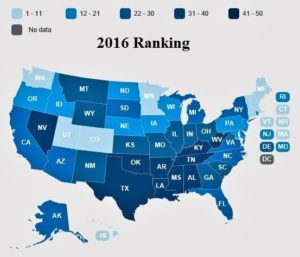
- Report: Nevada senior citizens’ health near bottom (pvtimes.com)2016 Senior Report, Overview (americashealthrankings.org)America's Senior Report Health Ranking, United Health Foundation (cdnfiles.americashealthrankings.org)
Nevada’s senior citizens’ health and quality of life were again ranked among the lowest nationally in a UnitedHealthcare report released last week, placing 42nd...America’s Health Rankings’ 2016 Senior Report takes into account 35 factors, including senior obesity and smoking rates, prevalence of falls, flu vaccination statistics and senior volunteerism...Nevada’s ranking was dragged down by factors including the state’s excessive drinking rate among seniors — self-reported at 9.2 percent...Nevada also suffers from comparatively low community support expenditures and flu vaccination coverage...The state’s strengths include a low prevalence of falls among seniors and a low rate of preventable hospitalizations among Medicare beneficiaries...Sandra Owens, a UNLV social work professor and expert in senior care, reviewed the report and described it as a good assessment of a state with work to do to improve health care for its elderly and overall populations.
- Pharmacy Week in Review: June 3, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- NGO questions drug industry backing of G7 antibiotic preservation pledge (in-pharmatechnologist.com)
Industry backing for a G7 plan to preserve antibiotics is at odds with current marketing practices says an NGO calling for a clamp down on campaigns that promote overuse...This week a pledge by G7 leaders to address 'market failures' that discourage antibiotic R&D and encourage over promotion won the support of the Association of the British Pharmaceutical Industry...The ABPI welcomed the ・commitment for collaborative global action by the G7 on tackling the unique medical, scientific and economic challenges presented by antimicrobial resistance・ telling us antibiotic stewardship was a core focus of the industry declaration launched at World Economic Forum meeting in Davos...industry support for efforts to preserve antibiotics is disingenuous according to Gemma Buckland Merrett, senior research Manager at NGO Health Action International, who suggested current marketing practices are part of the problem..."Evidence is starting to show that increased marketing of antibiotics by the pharmaceutical industry may be exacerbating inappropriate antibiotic use" Merrett told us...Despite the risks of resistance, pharmaceutical companies have continued to market antibiotics, often incentivising medical and veterinary personnel to overuse or inappropriately prescribe antibiotics.
- More Charges Against Ex-Pharmaceutical Executive Martin Shkreli (dddmag.com)
Federal prosecutors in New York have filed additional criminal charges against a pharmaceutical executive who separately was heavily criticized for raising the price of a lifesaving malaria medication...A new indictment filed Friday in Brooklyn says Martin Shkreli and his former attorney Evan Greebel schemed to defraud potential investors of his former drug company Retrophin Inc...They say the two allocated company stock to seven employees to conceal Shkreli's ownership of it...Shkreli previously pleaded not guilty to charges he lost investors' money through bad trades and looted the pharmaceutical company to pay them back.
- Independence of compliance reviews is questioned in drug firm settlements (reuters.com)
Some major U.S. drug companies have hired their own auditors to perform compliance reviews mandated in government settlements over alleged civil violations, such as paying kickbacks or off-label drug promotion, according to federal records...Third-party compliance reviews are playing an increasingly large role in helping the government ensure companies fulfill their obligations in federal settlements...Some lawmakers and legal experts say that practice creates conflicts of interest and could impair outside reviewers' independence...Susan Gillin, an attorney in the inspector general's office, said she agreed that "theoretically, a potential conflict of interest exists."...But she said her agency has not observed any problems with weak oversight by auditors serving in dual roles. Such arrangements are allowed, she said, as long as the firm is not involved in corporate management or advising on policies it will also review...Senate Judiciary Chairman Charles Grassley...said such arrangements could weaken enforcement..."If the firms enforcing the agreements aren't independent,"...that "defeats the purpose of the agreements."
- Heavyweight champion of the world (pharmatimes.com)
For more than 15 years NICE has punched above its weight internationally but as it comes under attack for its methodology, will its international clout suffer?...The bad headlines are back; on 20 May, the National Institute for Health and Care Excellence...came under fire from the media for its decision to bar Roche's Perjeta (pertuzumab) for breast cancer from the NHS...While patient groups and charities are worried about the effect on cancer patients in England, who may be denied treatment, there may also be a knock-on effect in other countries, which still look to NICE for guidance over their own reimbursement decisions...NICE International...advises countries from Ghana to Kazakhstan on HTA (health technology assessment) methodology and implementation...Yet, its international spread has come hand-in-hand with the growing disillusionment about NICE's decisions...An IMS Institute study in 2013 compared the reimbursement of cancer drugs in five countries that used cost per QALY (NICE's quality-adjusted life years) methodologies, including the UK, with five using broader methodologies, including Germany and US. It found that the cost per QALY countries reimbursed fewer cancer drugs, had slower access to those they did adopt, and generally performed poorer in terms of cancer outcomes. Moreover, it was not clear that they saved much money.