- Teaching old drugs new treatments (outsourcing-pharma.com)
Drugs currently being used to treat psychosis or depression may be able to treat emerging viruses, according to new research led by the University of Leeds...The researchers found that some common drugs were able to prevent a particular virus from infecting cells by blocking the ion channels that regulate potassium levels...drugs that were able to inhibit the potassium ion channels were effective against the Bunyamwera virus...which is increasing in prevalence in Mediterranean countries, and is endemic in Africa, the Middle East, and some Asian countries...There are many drugs targeting ion channels that are currently in use for a wide range of conditions. Our work shows that some of these might be suitable to treat virus infections...If existing drugs are confirmed to be effective against known members of a particular virus family, this opens up the possibility of using these 'off-the-shelf' treatments in a rapid response against dangerous new related virus strains that emerge...
- The Declining Revenues of GlaxoSmithKline’s Pharmaceutical Segment (finance.yahoo.com)
GlaxoSmithKline’s Pharmaceutical segment has declined substantially in 2015 due to the divestment of its oncology business to Novartis...The...segment reported revenues of $14.17 billion in 2015, compared to about $15.5 billion in 2014...revenues declined by 1% following lower sales of Seretide and Advair, partially offset by increased sales of HIV products Triumeq and Tivicay, and new pharmaceutical products...the...segment’s contribution to total revenues declined from 67.3% in 2014 to ~59.2%...GlaxoSmithKline’s Pharmaceuticals segment is classified into the following two franchises:
- HIV products are marketed under ViiV healthcare, a company with GSK as a major shareholder, while Pfizer and Shionogi are other shareholders. The company completed the acquisition of Bristol-Myers Squibb’s R&D HIV assets on February 22...HIV products reported growth of 54%...about $3.2 billion...partially offset by declining sales of Epzicom/Kivexa.
- Global pharmaceuticals - respiratory, cardiovascular, metabolic and urology, immuno-inflammation, and established products...key numbers:
- ...the respiratory franchise, the drugs Seretide and Advair are losing their market share to the generic competition...revenue decline of 7%
- ...the cardiovascular, metabolic, and urology franchise, the drugs Duodart and Jalyn have shown strong performances, while Avodart...is exposed to generic competition since October 2015...sales declined by 9%
- ...the immuno-inflammation franchise, Benlysta is driving growth...sales improved by ~24% while the franchise sales improved by 16%
- ...the established products franchise are losing their market share to generic competition...a revenue fall of 15%...due to lower sales across global markets.
- Other pharmaceuticals franchise include few key products...Augmentin, Relenza, Dermatology products, and rare disease products...revenues...declined by 4%...lower sales for Augmentin, dermatology, and rare disease products, partially offset by the strong performance of Relenza
- McKesson to acquire Rexall Health from Katz Group (drugstorenews.com)
McKesson announced...its agreement to acquire Rexall Health from Katz Group for $3 billion Canadian ($2.2 billion in U.S. dollars). The acquisition is expected to strengthen McKesson’s position in Canada’s pharmaceutical supply chain...McKesson intends to acquire Rexall Health’s business, including approximately 470 retail pharmacies. Rexall Health will help McKesson leverage its existing portfolio of assets to drive growth along the entire value chain...The acquisition of Rexall Health supports McKesson’s commitment to drive value in the industry by improving healthcare solutions delivered in the pharmacy; it enhances our ability to provide best-in-class pharmacy care through an expanded retail footprint...The transaction is subject to review under the Investment Canada Act and by the Competition Bureau of Canada...
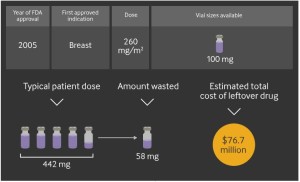
- Study: $3B will be wasted on unused portion of cancer drugs (cnbc.com)Overspending driven by oversized single dose vials of cancer drugs (bmj.com)
High prices for cancer medicines aren't the only reason they cost insurers and patients so much...Waste pads the bill…because infused cancer drugs are distributed in...vials that usually contain more medicine than most patients need. Most of the time that excess is thrown out, even though it's perfectly good — and worth hundreds or thousands of dollars...Researchers...estimate that wasted cancer medicine in the U.S. this year will add up to nearly $3 billion in excess costs...cancer medicines are one of the highest-priced, fastest-growing drug categories...study...in BMJ...details how drugmakers, hospitals and cancer doctors make money on unused cancer medicine. It focuses on the top 20 drugs for multiple cancer types packaged in single-dose vials and for which the dose depends on the patient's weight, finding that 1 percent to 33 percent of those 20 cancer drugs, on average, remains in vials after each dose is administered...The researchers say regulators could require manufacturers to supply multiple vial-size options, and the FDA and other federal health agencies need to reconcile their disparate guidelines on vial size and when leftover medicine from one vial can be given to another patient. Currently, sharing generally is only allowed within six hours of when the vial is opened to ensure the medicine's sterility.
- High Drug Prices Prompt Demands for Transparency (realclearhealth.com)
Outraged by exorbitant prices for certain prescription drugs, lawmakers in at least 11 states have introduced legislation that would require pharmaceutical companies to justify their prices by disclosing how much they spend on research, manufacturing and marketing...The sponsors of the measures say they have a variety of goals: to educate policymakers and consumers about the reason for high prescription drug prices; to shame pharmaceutical companies into moderating their prices; and, in some states, including Massachusetts, to actually place a ceiling on prices that are determined to be unjustified...A handful of transparency bills were filed last year, but none of them passed...The industry insists that it is being unfairly targeted by the transparency measures, pointing out that prescription drugs account for only 10 percent of health care spending in the United States, $300 billion out of $3 trillion per year...Some experts in drug pricing who are sympathetic to the goals of the transparency bills are dubious that they will accomplish what their sponsors hope...
- Europe launches new fast approval scheme for promising drugs (reuters.com)PRIME: priority medicines (ema.europa.eu)
European regulators launched a new scheme...to speed the approval of promising new drugs that address unmet medical needs by offering enhanced support to medicine developers as they work on clinical trials...The European Medicines Agency's initiative called PRIME, which stands for PRIority MEdicines, is the latest example of regulators on both sides of the Atlantic working to evaluate truly innovative drugs more swiftly than in the past...The European agency said it expected around 100 applications a year for its new PRIME scheme, which aims to foster better planning of medicine development to help companies generate the data needed for approval more rapidly...Experimental drugs accepted into the program will get continuous support from an EMA expert, who will be appointed early in the process and provide guidance on overall development plans and regulatory strategy.
- FDA still struggling with backlog of generic drug applications (statnews.com)
Under pressure to speed approval of generic medicines, the Food and Drug Administration...released data to defend its progress...the statistics indicate the agency is making headway, there are also clear signs the FDA continues to struggle with the workload...the number of full and tentative drug approvals has been rising each month since last April and reached 99 this past December...the agency also appears to be doing a better job of communicating with generic drug makers about their applications...Generic drug approval is gaining more attention thanks to the intensifying national debate over the rising cost of prescription medicines. Although prices have also risen for some of these copycat medicines, generics remain...lower-cost alternatives to brand-name drugs. And generics now account for 88 percent of all prescriptions written...The FDA is being a little disingenuous saying its backlog is almost cleared...The FDA faces...the increasingly large number of applications that drug makers are submitting...more than 4,000 have been filed in the past four years...the FDA workload will not abate...The upshot is that the rate at which new generics will find their way to pharmacy shelves is unclear — and that adds further uncertainty for health care budgets...
- Moody’s cuts pharma, medical tech sector outlooks to stable (cnbc.com)
Moody's Investors Service has reduced its outlook from positive to stable for both the pharmaceutical and medical device industries...The ratings agency...cited multiple headwinds, topped by two impacting both sectors. The strong dollar is reducing company sales outside the U.S., and insurers and other payers are pressing manufacturers to reduce prices in the U.S. and many other countries...Drugmakers also have been hurt by slower-than-expected uptake for many drugs anticipated to eventually produce annual sales in the billions, excepting a new generation of cancer drugs which stimulate the immune system. That's mainly because insurers and some foreign government health programs have been limiting or delaying patient access to new products that cost tens of thousands of dollars a year or more...Moody's...expects growth in earnings...will be 3 percent to 4 percent, down from 4 percent to 5 percent.
- Bass associate accuses PTO officials of bias against their pharma patent crusade (fiercepharma.com)
Hedge funder Kyle Bass and his Coalition for Affordable Drugs has been somewhat successful at using a new type of patent challenge to push review of "questionable" drug patents. But Bass' partner in that effort now says he doesn't trust the Patent & Trademark Office to be impartial in making those decisions...Erich Spangenberg, in an article at IPWatchdog, says PTO chief Michelle Lee and her staff have favored pharma since Bass announced last year that he and his group would target pharma patents via the inter partes review process...according to the documents, Lee and other PTO officials sat down with the industry association BIO...Bass and Spangenberg themselves called and emailed repeatedly to request a similar meeting, and they're still waiting... It's an outrage that Ms. Lee is showing incredible bias by granting meetings to the people that are gouging American patients, consumers and taxpayers and denying the same access to the people who are attempting to end this abuse...Some analysts have criticized the coalition for targeting inconsequential patents that aren't likely to change the length of a company's monopoly hold on the market, while profiting from shorting the shares of the same company. In a PTO filing, the coalition has acknowledged that Bass and his group don't have altruistic motives, but maintains that his profit motives "do not change the social value of his activities."...
- Endo to declare Opana ER addiction risks to settle opioid probe by NY (fiercepharma.com)
States across the country have been chasing painkiller makers, alleging all kinds of bad behavior in the sales and marketing of opioids. Now one of those cases has stuck, with Endo agreeing to not soft-pedal the risks of taking Opana ER (oxymorphone), or oversell its crush resistance as conditions of settling an investigation in New York...Endo Health Solutions also agreed to pay $200,000 and take a variety of steps to make sure doctors understand the addiction risks to their patients. The company will also stop marketing to and report any physicians they think might be improperly prescribing the opioid...the AG's (Attorney General Eric Schneiderman) investigation focused on Opana ER, which he said had become the drug of choice among many abusers...the investigation found that Endo "improperly marketed Opana ER as designed to be crush resistant," providing a "false sense of security to health care providers and their patients," even though Endo's own studies showed the drug could be crushed and ground up...