- Management Tools for Controlling Specialty Drug Costs (specialtypharmacytimes.com)
The increasing utilization and costs of specialty drugs will have a substantial impact on overall health care costs during the next decade...Pharmacy benefit manager tools that have successfully controlled costs in the traditional small-molecule drug categories will be critical to manage the increasing costs and requirements of the specialty drug category…
Financial Impact of Specialty Drugs: Current and Projected...by 2020, 9 of the 10 best-selling drugs (by revenue) will be specialty drugs...spending could reach $400 billion, or 9.1% of national health spending...Specialty benefit design and management tools employed by PBMs will also play a significant role in controlling specialty drug spending, as well as ensuring optimal patient care and support.
Specialty Benefit Design and Management...Specialty drugs frequently have unique shipping and storage needs, as a result specialty pharmacies are better equipped to procure, store, and dispense these treatments than traditional retail pharmacies. Furthermore, pharmacists and personnel at specialty pharmacies provide patient education and clinical support beyond the capabilities of a retail pharmacy...The tools in the PBM arsenal are critical to ensure appropriate care for patients needing specialty drugs while managing the often extraordinary costs.
Comprehensive management approaches that monitor and balance patient care outcomes and costs will help PBMs ensure that new, innovative medications are readily available and affordable to the patients who need them most.
- How drug ad narrators take the scariness out of side effects (statnews.com)
Drug makers are legally required to spend money telling you their products might lead to a series of horrifying side effects, up to and including death...Needless to say, for the pharmaceutical industry, this presents a communications challenge...But nearly two decades after television ads for prescription drugs began flooding American homes, drug makers have also perfected their delivery — allowing them to include the information they are obligated to provide while minimizing how scary it might sound... there’s an art to it...We use the same approach medical professionals do, telling a patient calmly: ‘We’re going to perform this surgery and there’s a 60 percent chance you won’t live,"...consumers absorb the most information when they can see people speaking rather than just hearing them. But "sometimes all the positives are said by someone you can see and all the negative things and risks are said by voiceover,"...Striking the right tone isn’t the only challenge for voice actors. Just think about making your living by rattling off phrases like "sphenopalatine ganglioneuralgia" or "vascular ischemia caused by thrombosis."..."You have to look up words a lot,"...
- U.K. cost gatekeepers say ‘show me’ to Alexion’s new rare disease med Kanuma (fiercepharma.com)
The U.K.'s cost-effectiveness gatekeepers think Alexion's newest rare-disease drug, Kanuma (sebelipase alfa), may be worth using in babies with lysosomal acid lipase deficiency. But it's going to need to see more research on the pricey med's costs and benefits before it shells out...At £491,992 ($704,630), the therapy doesn't represent good value for all patients, the National Institute of Heath and Care Excellence said in draft guidance. But there's still time for the company to change the watchdog's mind before it makes a final decision...Alexion...said in a statement that NICE had "failed to recognize the transformative clinical innovation of Kanuma" as a treatment for LAL deficiency, which can be particularly serious in babies. The inherited genetic disease causes fat to build up in cells, with survival for babies pegged at less than 12 months...it'll be up to the...drugmaker to prove the treatment's worth...
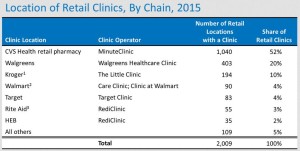
- Retail Pharmacy Clinics: Top Players and the Coming 2016 Pause (drugchannels.net)
As low-cost generics come to dominate retail dispensing activity, clinics have emerged as a way for drugstores and other retail pharmacy outlets to diversify into non-dispensing revenues...There are now about 2,000 retail clinics. Below, I examine the chains with the biggest market share: CVS Health, HEB, Kroger, Rite Aid, Target, Walgreens, and Walmart...In 2015, the number of clinics grew slowly. In 2016, I project another year of slow growth as the big players retrench...
- CVS Health’s MinuteClinic remains the largest and fastest-growing retail clinic business, now operating more than half of all retail clinics.
- Walgreens is the second-largest retail clinic operator, with more than 400 Healthcare Clinics (formerly Take Care clinics). The number of clinics in Walgreens retail stores declined in 2015.
- Kroger has emerged as the third-largest retail clinic operator with its Little Clinic business, which operates inside nearly 200 Kroger, Dillon’s, Fry's, JayC, and King Soopers stores.
- Rite Aid changed its clinic strategy with the 2014 acquisition of RediClinic. As of January 2016, Rite Aid operated 41 RediClinics....a further 35 retail locations.. in HEB grocery stores...At 14 Rite Aid locations, the company leases space to other clinic operators...
- Walmart launched its own Care Clinics in 2014. There are now 17 Care Clinics in Walmart stores...Walmart also leases space to independently owned and operated Clinic at Walmart operators...operate inside 73 Walmart stores.
- Q&A: CVS says this is how it’s lowering drug costs to consumers (statnews.com)
Rising prices for prescription medicines have become an increasingly contentious issue and cast a harsh spotlight on the pharmaceutical industry. As a result, a growing number of state and federal lawmakers are proposing legislation to somehow halt the trend. Drug makers maintain their prices fuel needed innovation and blame insurers for forcing consumers to pay higher costs. But Troy Brennan, the chief medical officer and executive vice president at CVS Health, argues differently. CVS runs the nation’s second-largest pharmacy benefits manager, which negotiates drug prices for companies and government agencies, among others. He maintains CVS is cutting costs to clients. Here is an excerpt of our conversation...
- Insurers, drugmakers wrestle with how to build value-based contracts (modernhealthcare.com)
...outcry over drug pricing has forced healthinsurers and pharmaceutical companies to devise ways of expanding access to potentially life-saving drugs without breaking the bank...Value-basedcontracts, in which insurers pay for drugs based on their effectiveness, have begun to sprout, and more are expected to follow. But experts believe these types of deals, although potentially beneficial, are not a panacea for managing drug costs. Value-based contracts may also be hard to orchestrate since they require doctors, insurers, pharmacy benefit managers and drug companies, often rivals, to cooperate and share data...What you still don't know at the end of the day with these outcomes agreements is whether they save money…Paying for the efficacy of drugs is a logical extension of how the U.S. wants to finance healthcare. Medicare rewards hospitals that deliver good care and penalizes those with high levels of costly readmissions. Private insurers have created narrow networks with selected health systems and doctors presumably based on their low costs and superior quality...most difficult hurdle may come once insurers, PBMs and drug companies have agreed on measurable, clinically relevant metrics. Those groups then have to share their medical and pharmacy data to track outcomes, which requires a high level of cooperation from fierce, often unfriendly negotiators...
- Teva Said to Seek Fast EU Approval for Allergan Unit Bid (bloomberg.com)
Teva Pharmaceutical Industries Ltd. will try to win an early European Union approval for its takeover of Allergan Plc’s generics unit by making concessions to regulators this week...Teva and Allergan will meet a deadline to propose remedies aimed at allaying any EU antitrust concerns...If EU regulators are satisfied with the proposals, they could clear the transaction without an extended probe that can last about 90 working days. The companies plan to close the deal by the end of March...Teva’s $40.5 billion cash-and-stock bid for Allergan’s generics drugs business will make it the world’s largest maker of generic medicines, giving it greater negotiating power with governments and private-health insurers. Allergan will receive a $1 billion termination fee from Teva if the deal fails to close due to regulatory issues.
- Ibuprofen patch heralds side effect free drug future (reuters.com)
An ibuprofen patch that delivers the painkiller directly through the skin to the site of pain, at a consistent dose for up to 24 hours, has been developed by UK researchers...Scientists at the University of Warwick and spin-off company Medherant say their TEPI patch could revolutionize the transdermal drug delivery market...the polymer matrix in the patch acts as a reservoir for the drug, capable of releasing between five and ten times the amount of drugs used in gels and patches currently on the market, for periods of six hours, 12 hours, and even 24 hours...The patches could help treat conditions like chronic back pain, neuralgia and arthritis without the need to take potentially damaging doses of the drug orally... the technology has exciting potential for other medications, such as opioid painkillers. What's important is to be able to extend the range of drugs that are available by patches because at the moment we're limited to about 20 different drugs, and there are thousands of drugs out there...which is limited by the technology of the adhesive...The TEPI patch incorporates new adhesive technology developed by...Bostik...Nutrition companies have also shown an interest in adapting Medherant's technology for transmitting minerals and vitamins through the skin.
- Toward saner drug pricing (washingtontimes.com)
If the FDA acts more quickly, lower costs and better patient outcome will follow...In a January hearing, Sen. Hatch queried Dr. Janet Woodcock, director of the Food and Drug Administrations’s Center for Drug Evaluation and Research, whether the agency’s backlog of some 3,500 lower-cost generic drugs awaiting approval was contributing to the cost consumers pay for drugs...Dr. Woodcock conceded that...slow-paced drug approvals contribute to high costs...Eighty-eight percent of all prescriptions in the U.S. are generics. If the FDA acted more quickly with both generics and new drugs, overall health costs would go down and patient health outcomes would improve...By quickly approving generics, lower cost versions would be available without unfairly infringing on profits (and thus stifling innovation) that pharmaceutical companies need to recoup the high costs to bring new drugs to market...Ms. Warren (Sen. Warren, Mass, Democrat) said...She’d like us to believe that Mr. Shkreli is representative of the broader industry and only government price controls can protect consumers from what appear to be an ever-rising cost of drugs...Rep. Stephen Lynch, Mass. Democrat, proposed what he amusingly called a "poison-pill amendment," which would allow Congress to decide when drug prices are too high, and then just eliminate patent exclusivity and "contract with DARPA — our government research labs — to produce your drugs at no cost to the consumer. That’s what we can do."...The Lynch and Warren approach will do one thing for sure. It will have a chilling effect on much needed private-sector investment in pharmaceutical research...the "market-failure, price control" argument would translate to higher overall treatment costs...
- WHO-backed pharmacovigilance program in India lags (fiercepharmaasia.com)
India's program to track the side effects of drugs in patients around the country is being hindered by low staff, lack of equipment and poor funding...India's pharmacovigilance program...currently sends data that medical professional collect of drug side effects to VigiBase--the database of the World Health Organization--as part of a global initiative to improve drug safety...Low staff numbers and poor equipment, as well as few properly trained personnel, have hampered the collection of this data...These important details about drug reactions could be dangerous to miss...The lack of quantity and quality in drug reaction data means India's government has less information on judging whether certain drugs have adverse side effects. Though the number of reports has risen...India has a long way to go to get their pharmacovigilance up to speed...