- French drug trial disaster leaves one brain dead, five injured (reuters.com) News Release (bial.com)
One person has been left brain dead and five others have been hospitalized after taking part in a clinical trial in France of an experimental drug made by Portuguese drug company Bial, French Health Minister Marisol Touraine said...90 people have taken part in the trial, taking some dosage of the drug aimed at tackling mood and anxiety issues, as well as movement coordination disorders linked to neurological issues...The six men aged 28 to 49 had been in good health until taking the oral medication at the Biotrial private facility that specializes in clinical trials..."This is unprecedented"..."We'll do everything to understand what happened."...The brain-dead volunteer was admitted to hospital on Monday...For three of the five others...there are fears of irreversible handicap...One of the six had no symptoms but was being carefully monitored...The medicine involved is a so-called FAAH inhibitor that works by targeting the body's endocannabinoid system, which is also responsible for the human response to cannabis...Bial said in a statement it was committed to ensuring the wellbeing of test participants and was working with authorities to discover the cause of the injuries, adding that the clinical trial have been approved by French regulators.
- BioMarin CEO on drug pricing, approval (video.cnbc.com)
BioMarin CEO Jean-Jacques Bienaime, discusses drug pricing, the sentiment of investors in biotech and his expectations for the company's Duchenne muscular dystrophy drug.
- Anthem Takes $3 Billion Express Scripts Fight Public (bloomberg.com)
Health insurer Anthem Inc. wants $3 billion a year more in savings on drugs from Express Scripts Holding Co., and is threatening to ditch the company in a move that would depose the pharmacy benefit manager as the country’s biggest...The insurer, which contracts with Express Scripts to manage prescription drug costs for its members, said the pharmacy manager should be passing along about $3 billion a year more in the savings it negotiates from drug companies... The two may be running out of time. “We have a very involved dispute resolution process in the contract that has been fully exhausted”...Anthem took the dispute public because the company wasn’t getting the savings it needed to offer more competitive products, such as Medicare drug plans..."Both of us have to step back and see whether we’re honoring the contractual terms of the agreement"...If not, “you have your legal remedies.”
- Health Care in 2016: Eight Charts You Need to Follow the Sector (bloomberg.com)
Next (this) week bankers, investors, companies and researchers from around the world will gather in San Francisco for the J.P. Morgan Healthcare Conference, a massive gathering that sets the tone for the rest of the year. These are the charts that will help you keep up with the industry in 2016.
- Remember, You've had It Pretty Good
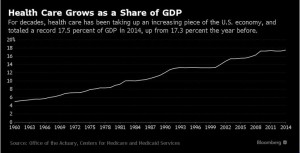
- Health Care Grows as a Share of GDP
- Are Obamacare's Gains Petering Out?
- Biotech, Insurers, Hospitals and Pharma All Gain Since 2010
- Venture Capital Investment Is at a Record
- Boom Years for Biotech
- The IPO Class of 2015
- Health Care's Big Deals
- Bayer opens large China plant for traditional Chinese and Western OTC meds (fiercepharmamanufacturing.com)
Bayer boosted the reach of its international consumer health business when it picked up Chinese herbal and OTC special Dihon in 2014. Now it has boosted its production capacity with a new plant that will manufacture both Western OTC products and traditional Chinese medicines (TCM)...The German drugmaker last week opened the first phase of the 1.4 billion yuan ($213 million) plant in Majinpu in the Yunan province of China. At 111,534 square meters (1,200,542 square feet), it is Bayer's second largest over-the-counter products manufacturing site in the Asia-Pacific... first phase of construction has secured GMP approval for part of the TCM production line...additional work is ongoing that will allow the company to start manufacturing Bayer's key TCM product, Dan E Fu Kang, which is marketed as a gynecological medicine for women's health indications including dysmenorrhea...
- Will GSK’s no-speaker-fee system work? Execs say yes; critics, not so much (fiercepharmamarketing.com)
GlaxoSmithKline has taken some bold steps to polish its image, tarnished by a Chinese bribery scandal and $3 billion settlement with the U.S. government. But naysayers blame its first big moves--nixing sales-rep quotas and pegging bonuses to "softer" measures instead--for disappointing roll-outs for several new meds...Now that it's dropping the time-tested tactic of paying doctors to promote its meds, critics are piling on there, too. But the U.K.-based drugmaker is sticking to its guns, recruiting its own doctors and other experts to tout its meds...Society now sees pharma-paid doctors as "hired guns,"...The only way to avoid that rap is for drugmakers to use their own employees instead...Since GSK first announced its no-payment policy--which went into effect Jan. 1--other Big Pharmas have addressed the question. Will they do the same? Several companies are on record with an unequivocal "No," saying that their doctor-speakers are important to their promotional efforts...Time will tell whether GSK's new policy will boost its credibility without cutting into revenue. More time will tell whether a buffed-up image would translate into bigger sales.
- Drug Makers Dismiss Outrage over High Prices as ‘Abomination’ (realclearhealth.com)
The protesters carried handwritten signs accusing drug maker Gilead Sciences of greed for pricing its breakthrough hepatitis C drug at $84,000 per treatment...a Gilead executive was asked how he lives with himself...the executive vice president for corporate and medical affairs, joked that he goes running...the drug industry’s biggest showcase, the J.P. Morgan Healthcare conference...As executives and investors shuttled from meeting to meeting, seeking deals, many dismissed public outrage at the industry as misguided...Public anger at drug companies is “an abomination,” Ron Cohen, chairman of the big industry group BIO, said at the Biotech Showcase...All the talk about pharma profiteering, Cohen said, is “a perversion of reality.”...Outside the stately old hotel where the...conference is being held, a handful of protesters marched with signs declaring “Gilead = Greed,” “Public Health Not Private Wealth,” and “Jail Gilead Drug Profiteers.”...“If Gilead’s approach is the future of how blockbuster drugs are launched in America, it’s going to cost billions and billions of dollars to treat just a fraction of patients in America,” Senator Ron Wyden said...
- State measures to slash drug prices face big hurdles (statnews.com)
When shopping for a good deal, people usually want the same bargain as the next guy. And a group of AIDS activists is taking this approach in hopes of containing the rising cost of prescription drugs...In California and Ohio, they’re pushing ballot measures that would require state programs — such as Medicaid or prison systems — to pay no more for medicines than the Department of Veteran Affairs... “The goal [ballot initiatives] is to obtain lower prices, as well as gain more transparency on industry pricing, which is opaque,” said Ged Kenslea of the AIDS Healthcare Foundation...“And we’re hoping these will serve as a catalyst to other organizations or state legislators to enact similar measures in other states.”...well-intentioned, the effort may be somewhat quixotic...“These state initiatives might save a fistful of money in the short term and the first states where they are passed will get most of the benefit,”...“But the companies will find a way to compensate. So it isn’t likely to solve the problem in the long run.”...Still, the state initiatives may yet lead to change, if only because a groundswell of citizen-driven ballot measures might just force lawmakers to pay attention. In the end, getting a better deal might only be a vote away.
- FDA castigates China’s Zhejiang Hisun for ‘systemic data manipulation’ (fiercepharmamanufacturing.com)
A Chinese drugmaker that has a joint venture with Pfizer to produce generic drugs and plans to separately make biosimilars, has been savaged by the FDA in a warning letter for serious data manipulation and shipping to the U.S. products that repeatedly failed follow-up testing by customers...A warning letter posted by the FDA today shows why the agency last September decided to ban products coming out of the Zhejiang Hisun Pharmaceutical plant in Taizhou City. "We observed systemic data manipulation across your facility, including actions taken by multiple analysts, on multiple pieces of testing equipment, and for multiple drugs,"...its concerns over data manipulation were "heightened by the significant number of customer complaints for subpotency and out-of-specification impurity levels the company received from 2012-2014." There were more than 60 complaints for impurity problems...The FDA said the explanations that Zhejiang Hisun Pharmaceutical gave for why data was deleted and raw data was no longer available were completely inadequate, as were the responses to other issues. It said it will continue to prevent the company's products from coming into the U.S. until the agency is satisfied with the steps the drugmaker has taken to bring up its manufacturing standards and can convince the FDA that its test data is authentic.
- Drug Price Policy Can’t Discourage R&D: Biogen CEO (bloomberg.com)
George Scangos, chief executive officer at Biogen, talks with Betty Liu about the company's drug pipeline and explains the complications related to drug pricing and research and development. He speaks on "Bloomberg Markets." (Source: Bloomberg)