- State, Local Officials Push Manufacturers to Pay for Drug Disposal (realclearhealth.com)
...drug disposal is expensive — the Nebraska program spends $10 per pound to ship and destroy medication — and some states and municipalities want drug companies to pick up the tab…Six municipalities in California require drugmakers to pay for take-backs. Nine states...have considered similar measures...Proponents say these laws, which are similar to programs that require manufacturers to pay for electronics recycling, would make it easier for patients to dispose of prescriptions. But drugmakers oppose the local mandates and warn that disposal costs could be passed on to patients through higher drug prices…Alameda County in California was the first jurisdiction to pass a law requiring manufacturers to pay for take-backs, in 2012, though the industry was quick to file suit, arguing it violated the interstate commerce clause of the U.S. Constitution. The Alameda law was upheld this year when the Supreme Court refused to hear the industry’s appeal…Drugmakers would rather lead their own take-back programs than comply with local or state mandates, said Priscilla VanderVeer, of the Pharmaceutical Research and Manufacturers of America...Because drugmakers sell across the country, meeting the customized disposal requirements of local programs is complicated...There are also more effective ways to get rid of the medicines, she said, like at-home disposal or returning unwanted drugs using special envelopes with carbon lining…“That’s just a financial and logistical nightmare for something that’s not necessarily going to be effective, or secure or environmentally helpful,” she said.
- Advising Students on Business Plans (pharmacytimes.com)
Bryan Ziegler, PharmD, executive director of Kennedy Pharmacy Innovation Center, talks about what his experience has been like as an adviser for the South Carolina College of Pharmacy student business plan team.
- Publix enhances free medication program (chaindrugreview.com)
Publix Super Markets Inc. has expanded supply options in its free medication program to help patients on maintenance medicines…The food and drug retailer said...that Publix Pharmacy will now offer free 90-day supplies of metformin, lisinopril and amlodipine to customers with a prescription for the medications. Previously, 30-day supplies of these medications were offered under the retailer’s free medication program… Publix noted that it enhanced the program to promote its pharmacy patients’ medication adherence…“Publix Pharmacy continues to enhance our free medication programs to best serve the needs of our customers,”… “With health care and prescription costs continuing to rise, medication adherence may prove challenging. The enhancement to our free prescription programs and pharmacy services allows our customers to focus on staying healthy and achieving their overall wellness goals.”
- New OIG Report Shows Hospitals’ Huge 340B Profits from Medicare-Paid Cancer Drugs (drugchannels.net)Part B Payments for 340B-Purchased Drugs (oig.hhs.gov)
..Office of Inspector General (OIG) has just released another eye-opening report on the 340B drug discount program: Part B Payments For 340B-Purchased Drugs.…The report documents how 340B-eligible hospital outpatient departments earn tremendous profits from the Medicare Part B program…OIG focuses on how the Medicare program could save money by sharing in these mega-profits. As usual, we don’t know if needy patients are benefiting from hospitals’ excess Medicare 340B profits. The OIG even reminds us three times in its report: “The 340B statute does not restrict how covered entities may use these funds.” Unfortunately, the Health Resources and Services Administration’s recent Omnibus Guidance didn’t bother to require that hospitals use 340B funds help the neediest patients access valuable medicines. Money, so they say, is the root of all evil today.
- E-mails reveal concerns about Theranos’s FDA compliance date back years (washingtonpost.com)Surprise: Theranos CEO Says Company Is Doing More Tests Than Ever (forbes.com)A comprehensive guide to Theranos’s troubles and what it means for you (washingtonpost.com)Hot Startup Theranos Has Struggled With Its Blood-Test Technology (req sub) (wsj.com)
Years before Theranos…came under harsh public scrutiny in October, a military official raised concerns that the secretive company was violating federal law…E-mail correspondence obtained by the Post reveals that an official evaluating Theranos’s signature blood-testing technology for the Department of Defense sounded the alarm in 2012 and launched a formal inquiry with the Food and Drug Administration about the company’s intent to distribute its tests without FDA clearance — a problem that has resurfaced this year, leading Theranos to temporarily stop offering almost all of its tests...In October, a Wall Street Journal investigation revealed the company was running most of its tests on devices made by other firms instead of its own “breakthrough” technology. The Journal reported that former Theranos employees and executives of Safeway had questioned the accuracy of the tests.
- Pharmacy Week in Review: December 4, 2015 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program highlights the latest in pharmacy news, product news, and more. (video)
- FDA delays proposed rule on safety warnings for generic drugs again (statnews.com)Supplemental Applications Proposing Labeling Changes for Approved Drugs and Biological Products (federalregister.gov)
For the second time, the Food and Drug Administration has delayed a controversial rule for updating generic drug labeling that would allow generic drug makers to independently update safety warnings, something only brand-name drug makers can currently do before receiving regulatory permission…the agency said the rule will be published in July 2016…The delay came amid push-back from the pharmaceutical industry, as well as some congressional lawmakers who questioned whether the agency has the authority to issue such a rule…FDA said it wants to “create parity” between brand-name and generic drug makers…generic drug makers have not been required to run tests to prove their medicines are the same as brand- name drugs and, therefore, have not had to upgrade labels with safety information…the generic trade group has argued the FDA rule could add $4 billion to the nation’s health care bill because regulatory requirements and litigation costs would eventually force manufacturers to raise prices.
- National Health Spending In 2014: Faster Growth Driven By Coverage Expansion And Prescription Drug Spending (content.healthaffairs.org)
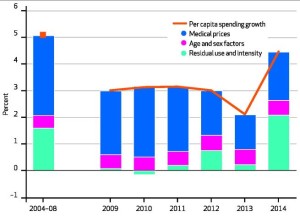
Factors Accounting For Growth In Per Capita National Health Expenditures, Selected Calendar Years 2004–14
US health care spending increased 5.3 percent to $3.0 trillion in 2014. On a per capita basis, health spending was $9,523 in 2014, an increase of 4.5 percent from 2013. The share of gross domestic product devoted to health care spending was 17.5 percent, up from 17.3 percent in 2013. The faster growth in 2014 that followed five consecutive years of historically low growth was primarily due to the major coverage expansions under the Affordable Care Act, particularly for Medicaid and private health insurance, which contributed to an increase in the insured share of the population…growth in total retail prescription drug expenditures accelerated sharply, increasing 12.2 percent to $297.7 billion…The strong growth in prescription drug expenditures in 2014 was caused by increased spending on new medicines, a smaller impact from patent expirations than in previous years, and price increases for brand-name drugs. The single largest driver of growth in specialty drug spending in 2014 was the impact of new treatments for hepatitis C, which contributed $11.3 billion in new spending.
The Affordable Care Act | Factors Accounting For Growth | Revisions To The National Health Expenditure Accounts | Sponsors Of Health Care | Private Health Insurance | Medicaid | Medicare | Out-Of-Pocket Spending | Retail Prescription Drugs | Hospital Care | Physician And Clinical Services
- Prisons can lock up savings by treating inmates with hepatitis C (statnews.com)
Here’s a chance to lock up some savings…The high cost of hepatitis C drugs may be straining budgets nationwide, but the country can save money if prison inmates are screened and treated for the disease… researchers determined that treatment could lower infection among both prisoners and society…If more inmates are treated, the disease would be less likely to spread within prisons, where an estimated 17 percent of the population is already infected. Beyond that, hepatitis C would be less likely to spread in the wider population once inmates are released. About 1 percent of the general US population is believed to have hepatitis C…screening and treatment would require an added 12.4 percent increase in the health care budgets of state and federal prisons during the first year such an effort is undertaken. But that would eventually decline to an annual budget increase of less than 0.7 percent after 15 years…prices have prompted many public and private payers to restrict access to treatment…a...class action lawsuit was filed earlier this year by two inmates in Massachusetts state prisons for failing to provide hepatitis C treatment to most infected prisoners…
- GPhA speaks out on generics labeling, access (drugstorenews.com)
…the Generic Pharmaceutical Association released statements related to the current state of generics, specifically proposed changes to rules regulating labels on generic drugs from the Food and Drug Administration, as well as steps it believe lawmakers should take to ensure affordable medication access…
- …accessibility, the organization recommended that lawmakers should encourage the FDA to review the approximately 3,800 generic drug applications awaiting action in a timely manner.
- …wider use of generics among low-income Medicare beneficiaries, which it estimates could save $17.7 billion over the course of 10 years.
- …passing the FAST Generics Act, which it says will keep drug companies from using Risk Evaluation and Mitigation Strategies to keep generics from hitting the market…
- …called for a repeal of a Medicaid rebate increase for generic drugs included in this year’s budget agreement.
- … regulations concerning labeling of generics, as the organization says currently, branded and generic drugs have the same label, but a proposed change to generic labeling requirements would require manufacturers to update labels without frist (sic) getting FDA approval…proposed the Expedited Agency Review (EAR), which relies on the FDA to review new safety information and take action on label changes.
- … EAR suggests time parameters for the FDA to take action and encourages the adoption of e-labeling for real time information sharing rather than continuing the reliance on paper label changes that take months or years to adopt.









