- Why Inversion Deals Are All the Rage in Health Care (bloomberg.com)
Marshall Sonenshine, Sonenshine Partners chairman, comments on Pfizer possibly being in advanced talks to buy Allergan for as much as $380 per share. He speaks with Bloomberg's Betty Liu on "Bloomberg Markets."
- Apixio launches cognitive computing platform (healthcareitnews.com)
Apixio announced…the release of its new cognitive computing platform, Iris, which it says will bring advanced data insights to healthcare by extracting and analyzing medical data previously trapped in electronic health records…The U.S. annually produces 1.2 billion clinical care documents, but about 80 percent of the data is unstructured and difficult to access…"Making sense of unstructured healthcare data is extremely challenging and requires sophisticated technology like cognitive computing to make the information useful,"…Iris is meant to give healthcare institutions access to patient data to create a more accurate care profile, thus improving the quality and efficiency...
- Feds call on hospitals to review disaster plans after Paris attacks (fiercehealthcare.com)
French hospitals' mass casualty response can be a guide for United States..The horrifying attacks on restaurants, clubs and cafés in Paris last week have resulted in a call by U.S. federal agencies for healthcare providers and hospitals to review their disaster plans and to exercise "enhanced vigilance"…Department of Homeland Security and Health and Human Services urged healthcare providers this week to "review and exercise their security plans" in the wake of the attacks…The notice urged healthcare organizations to:
- Review security plans and conduct drills that incorporate scenarios based on tactics recently used
- Review active shooter, suspicious activity reporting, and counter-IED preparedness in training and awareness initiatives and in organizational safety briefings
- Ensure proper functioning of emergency communications equipment and conduct regular tests
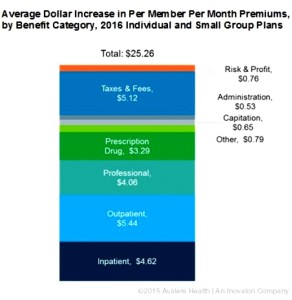
- Despite rhetoric, new data shows medicines are not primary driver of premium increases (catalyst.phrma.org)
Despite claims from insurers about the impact of medicine costs on premium increases, their own data indicates otherwise. In fact, just $3.29 of the average $25.26 increase in monthly premiums in 2016 is due to prescription drug costs, according to new research released yesterday by Avalere Health. So what is driving premium increases? Avalere found that the largest driver of premium increases was hospital services…analysis is based on actuarially-certified analysis submitted by the plans themselves to justify their premium rate increases. This data shows that prescription drugs dispensed at a pharmacy represent a smaller share of premium increases than inpatient hospitalization, outpatient hospitalization, professional services or taxes and fees. In fact, even if plans did not anticipate any increase in prescription drug spending, the average premium would still increase by more than $20 per month.
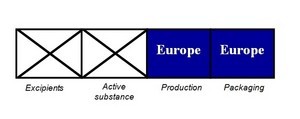
- French proposal for ‘Made in EU’ labels threatens to divide drug industry (statnews.com)Decree on the specification of the place of manufacture on the external packaging of pharmaceutical products (ec.europa.eu)
In a controversial move, France has asked the European Commission to allow new labeling on medicines that would indicate whether products and ingredients come from Europe or elsewhere. But the effort is likely to divide drug makers amid concerns about the quality of prescription drugs…The stated reason for the request is to bolster transparency, because French consumers may have “doubts” about their medicines, according to the filing by the French government. And the notice singled out generic medicines, in particular…The filing also follows the recent suspension of some 700 mostly generic drugs by the European Union. The EU acted after French regulators questioned the reliability of clinical trial data generated by GVK Biosciences, a contract research organization in India that conducts studies for drug makers.
- FDA approves Adapt Pharma’s nasal spray for opioid overdose treatment (reuters.com)
Food and Drug Administration approved the first-ever nasal spray emergency treatment for opioid overdose…The spray, developed by privately held Adapt Pharma Ltd, uses naloxone, a drug used to treat opioid overdose for nearly 45 years but approved only in injectable forms…The treatment…is expected to have wide coverage under health insurance with affordable co-pays…Group purchasers, such as law enforcement, fire fighters, departments of health, local school districts, colleges and universities, and community-based organizations will be able to purchase the spray at a discounted price of $37.50 per 4 mg device…
- Price controls loom for generic drugs in India next year (fiercepharmaasia.com)
India's generic drugs could face new price controls as early as next year, as the prime minister's office pushes for an investigation of what one official in press reports called "astronomical" markups by drugmakers, suppliers and retailers…The office reportedly ordered the Department of Pharmaceuticals to look into the large markups that occur on generics that are sold through distributors…a panel already has been established to include the department, the National Pharmaceutical Pricing Authority and the Competition Commission of India, as well as industry groups…For several years, the government has attempted other ways to make generics more affordable to Indians, including the establishment of its own drug store chain, Jan Aushadhi. By the end of the year, the group expects to have more than 400 generics for sale, compared with the 225 now available. Within two years, the government expects to have 3,000 Jan Aushadhi stores throughout the country.
- EPA Seeks Input on Drug Flushing Ban (hhnmag.com)
All health care facilities would be banned from flushing drugs among other changes in a proposed rule…Stop flushing pills down the toilet…That’s the message behind a proposed rule from the Environmental Protection Agency that would ban health care facilities from disposing of hazardous pharmaceuticals by flushing them…significant changes may be required if the rule is enacted…The EPA estimates that the proposal is projected to prevent the flushing of more than 6,400 tons of hazardous waste pharmaceuticals annually…The rule would fall under the Resource Conservation and Recovery Act, and is open for comment from the public until Dec. 24.
- India’s Supreme Court wants stricter oversight of MSD, GSK HPV clinical trials (fiercepharmaasia.com)
Suggesting that India's chief drug regulator may not be doing enough to protect certain volunteers in clinical trials, the Supreme Court ordered the agency to provide adequate oversight, particularly when drugs to treat the human papillomavirus are concerned…Central to the high court's concerns, it said, were Merck Sharpe & Dohme's Gardasil and GlaxoSmithKline's Cervatrix...The two justices presiding said they were not trying to run the Drugs Controller General of India, but they believed the poverty-stricken and others in trials should be protected at the informed-consent stage when testing HPV vaccines… The court said the procedure should be transparent and patients should be compensated when failures occur.
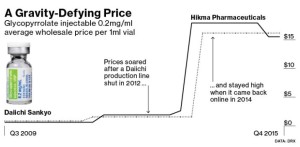
- Broken Market for Old Drugs Means Price Spikes Are Here to Stay (bloomberg.com)
Prices for 50 hospital drugs have risen 10-fold over 8 years…Generic injection meds in short supply as producers drop out…With most products, you’d expect a flood of new supply to quickly drive back down a price spike caused by a temporary shortage. Not so in the topsy-turvy world of hospital pharmaceuticals…All sorts of medications, from treatments for irregular heartbeats to supplies as basic as saline solution, have shot up in price as common hospital drugs are increasingly, and sometimes critically, in short supply. The reasons for the shortages are varied: creaky old factories, an FDA crackdown on quality control, companies exiting the market for more lucrative opportunities and fewer producers in the wake of a wave of mergers and acquisitions…“It’s a broken market," said Stephen Schondelmeyer, a pharmacist and economist at the University of Minnesota who has studied drug prices. "Drug companies know there is going to be an end to this blank check era and they are pushing for whatever they can get."