- Independent Pharmacy Economics: Profits Steady, but Sales Down (Maybe) (drugchannels.net)
Time for my annual look at independent pharmacy owners’ business economics, drawn from the recently released 2015 National Community Pharmacists Association Digest…The data reveal that independent pharmacy owners are doing better than you might expect…The number of independent pharmacies continues to hold steady…In the NCPA Digest sample, average per-prescription revenue declined, which reduced gross profits and average prescription revenues. On the other hand, IMS Health data imply independents' revenue and prescriptions are growing, not declining. Hmmm.
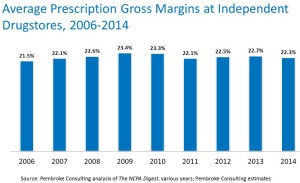
- Overall independent pharmacy profit margins remain stable.
- An independent pharmacy’s prescription profit margins were also stable.
- An independent pharmacy’s gross profit per prescription declined.
- Brand-to-generic substitution is reducing retail pharmacy revenues.
- The average pharmacist owning a single pharmacy earned about $228,000 in 2014.
- The total number of independent pharmacies continues to hold steady.
- Global drug spending to hit $1.4 trillion in 2020: IMS (reuters.com)
Global spending on medicines will reach $1.4 trillion in 2020, driven by increased healthcare access in emerging markets and high-priced new drugs for cancer and other diseases…That is up from about $1.07 trillion this year, representing a compound annual growth rate of 4 to 7 percent over the next five years…Some 225 new drugs will come to market over the next five years, about a third aimed at cancer, as well as medicines for rare diseases, which can carry ultra-high price tags, and treatments for autoimmune diseases and heart disease…cost will be partially offset by patent expirations expected to reduce spending on branded medicines by $178 billion, including $41 billion from biologic drugs as cheaper biosimilars become more widely adopted…Developed markets will still account for the lion's share of global spending, about 63 percent, due to higher prices and access to the newest, most expensive treatments.
- Suburban Detroit Doctor Receives 7 Year Sentence for Writing Unlawful Prescription for Controlled Substances including Oxycodone (dea.gov)
Hussein “Sam” Arwada also admitted to defrauding the health care system of $2.3 million…physician who practiced in Warren, Michigan, was sentenced to 84 months in prison today for writing prescriptions for oxycodone and other controlled medications without medical justification, and for health care fraud…he conspired with James Lyons, a patient “marketer,” and others, to write prescriptions for 80,000 doses of oxycodone, and other controlled medications. The prescriptions were written in the names of people who were brought to him by Lyons and other marketers, for no legitimate medical purpose. The marketers then bought the pills from the “patients” and re-sold them to street dealers. Awada then used the patient data...to submit bills to Medicare and Blue Cross Blue Shield for services that were either never performed or were medically unjustified. Awada caused these same patients to receive monthly x-rays, and other invasive tests, which were medically unnecessary but helped to conceal his fraud.
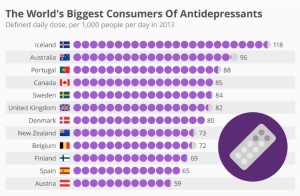
- The World’s Biggest Consumers Of Antidepressants (forbes.com)
Iceland is the biggest consumer of antidepressants worldwide, according to recent OECD (Organisation for Economic Cooperation and Development) report entitled “Health at a Glance 2015.” Some 118 out of every 1,000 Icelanders now consume these drugs on a daily basis, though the trend certainly isn’t new...Some experts attribute the country’s use of these drugs to a weakening of social taboos, along with a greater tendency to seek treatment. Some also believe there is a link between antidepressant consumption and the failure of all three of Iceland’s main banks during the financial crisis. Australia is in second position with 96 out of every 1,000 people taking antidepressants daily, while Portugal rounds off the top three with 88 per 1,000.
- We have met the enemy, and he is us (drugtopics.modernmedicine.com)
The Cynical Pharmacist lays it on the line..
- Back in the day - Not long ago, pharmacists were respected. We went out of our way to help people in every imaginable situation…What is the upshot of all this kindness today? Patients berate and complain about us unless or until we accede to their every demand.
- Think about what you’re doing…If a prescriber forgets anything on a prescription, what do pharmacists do? We call.
- Whose job is it?...What do we do? What should we do?...What happens when I send prescriptions back with a patient?
- Do the rules apply?...prescribers as well as pharmacists. I believe that both groups of people have to read, understand, and follow the same set of rules…Somehow it has become our job to fill in the blanks for the prescribers…How about a big fat “no”?
- Who ends up on the hook?...But what about the poor patients? Don’t put them in the middle of a pissing contest between you and the prescriber. It’s not their fault their prescriber was in a hurry/forgot/is a moron.”
- Stand up for pharmacy…I’ve done it. It’s amazing how quickly physician offices change their tune when facing legal repercussions.
- Just stop…we are great complainers. We also shy away from conflict. Patients yell at and abuse us all day long and we complain — after they’ve gone…Stop permitting abuse.
Better yet…just send them back.
- Physicians call for fairness in drug prices, availability (ama-assn.org)
In response to increasing drug costs impacting patient access to needed medications, physicians voted...to convene a task force and launch an advocacy campaign to drive solutions and help make prescription drugs more affordable. Physicians also are calling for greater competition in the pharmaceutical industry and transparency in prescription drug prices and costs… The new policy calls for the AMA to generate an advocacy campaign to engage physicians and patients in local and national advocacy initiatives to bring attention to the rising price of prescription drugs and put forth solutions to make them more affordable for all patients…Actions called for in the new policy include:
- Support legislation to shorten the exclusivity period for biologics.
- Encourage actions by federal regulators to limit anticompetitive behavior by pharmaceutical companies attempting to reduce competition from generic manufacturers through manipulation of patent protections and abuse of regulatory exclusivity incentives.
- Encourage prescription drug price and cost transparency among pharmaceutical companies, pharmacy benefit managers and health insurance companies, which will help patients, physicians and other stakeholders understand how drug manufacturers set prices, and the prescription drug tiering and cost-sharing requirements of health plans.
- Monitor pharmaceutical company mergers and acquisitions in the pharmaceutical industry.
- Support a balance between incentives for innovation and efforts to reduce regulatory and statutory barriers to competition as the patent system is evaluated and potentially reformed.
- Southern Hills hospital maintains top performing status (reviewjournal.com)More than 1,000 hospitals named Joint Commission 'top performers' (fiercehealthcare.com)
For the fifth consecutive year, Southern Hills Medical Center in Las Vegas has made the list of top performing hospitals compiled by the independent agency that accredits the most U.S. health care organizations…Fourteen of the 27 Nevada hospitals that submitted data last year to The Joint Commission attained top performing status, including seven other facilities in Southern Nevada. In addition to Southern Hills, the commission also recognized Sunrise, Mountainview, Valley and Desert Springs hospitals in Las Vegas; Strategic Behavioral Health's Red Rock facility; the Spring Mountain Treatment Center in Las Vegas; and Mesa View Regional Hospital in Mesquite.
- Why FDA Should Oversee Laboratory Developed Tests (blogs.fda.gov)The Public Health Evidence for FDA Oversight of Laboratory Developed Tests: 20 Case Studies (fda.gov)Framework for Regulatory Oversight of Laboratory Developed Tests (LDTs) DRAFT GUIDANCE (fda.gov)Theranos isn’t the only diagnostics company exploiting regulatory loopholes (theverge.com)
Today FDA is issuing a report that illustrates the real and potential harms to patients and to public health from certain laboratory developed tests (LDT) – tests that are designed, manufactured and used in a single laboratory…But times have changed. LDTs have increased in complexity and availability and are now frequently used to diagnose common, serious medical conditions, including cancer and heart disease, with potentially greater impact on patients...LDTs are still under a general policy of enforcement discretion. That means they have rarely undergone FDA review to determine whether they are accurate, reliable, and provide clinically meaningful results...FDA’s own adverse event reporting databases rarely capture problems associated with a faulty LDT...the Agency was able to pull together 20 case studies based on information available in the public domain that show how lack of LDT oversight may be causing or is causing significant harm to patients…FDA has proposed to step up our oversight of LDTs. We issued a draft guidance last year which we’re currently working to finalize, that proposes to phase in enforcement of premarket review requirements for LDTs. FDA oversight would help ensure that tests are supported by rigorous evidence, that patients and health care providers can have confidence in the test results, and that LDTs have more scientifically accurate product labeling.
- Valeant faces new trouble as dermatologists sour on its drugs (statnews.com)

FILE PHOTO. The head offices of Valeant Pharmaceutical is pictured in Montreal on Monday May 27, 2013. THE CANADIAN PRESS/Ryan Remiorz
Yet another sign of trouble for Valeant Pharmaceuticals has emerged. The beleaguered drug maker already faces congressional hearings into its pricing practices. Now, one of its bread-and-butter product lines may not ring the register as often as before…Fewer dermatologists are prescribing Valeant medicines…The change constitutes more fallout from a scandal involving the drug maker’s relationship with a mail-order pharmacy known as Philidor Rx Services. Valeant reportedly hid its ties to the pharmacy in order to inappropriately boost prescriptions and insurance reimbursements...68 percent are writing fewer prescriptions for Valeant’s dermatology products and a similar percentage expect they will not prescribe Valeant drugs in coming months.
- American Medical Association wants to ban drug ads to consumers (statnews.com)AMA Calls for Ban on Direct to Consumer Advertising of Prescription Drugs and Medical Devices (ama-assn.org)
American Medical Association is calling for a ban on advertising prescription drugs and medical devices directly to consumers. The move…is largely symbolic, because any ban would have to be authorized by Congress…The new AMA policy comes after years of complaints by physicians…some ads too often encourage patients to seek medicines unnecessarily. They also resent the pressure the ads place on them to write prescriptions out of concern patients will switch physicians…Another rationale for the ban, however, is the rising cost of drugs…many of the ads aimed directly at consumers promote more expensive medicines…the Pharmaceutical Research and Manufacturers of America…“Providing scientifically accurate information to patients so that they are better informed about their health care and treatment options is the goal of direct-to-consumer pharmaceutical advertising...“Beyond increasing patient awareness of disease and available treatments, DTC advertising has been found to…encourages patients to visit their doctors’ offices for important doctor-patient conversations about health that might otherwise not take place.”...