- Brexit Is Seen Costing Pharma Hundreds of Millions of Pounds (bloomberg.com)
Brexit uncertainty is forcing drugmakers in Britain to invest hundreds of millions of pounds to maintain the steady supply of medicines to patients, siphoning off money that could have gone to developing new treatments...Pharmaceutical companies are preparing for potential regulatory and trade hurdles after the U.K. leaves the European Union, expanding testing facilities, moving marketing authorizations and drafting contingency plans. Regulatory alignment between the EU and Britain makes sense to all sides, and if Britain remains part of Europe’s drug-approval system, a lot of the investment may prove to have been unnecessary, said Mike Thompson, chief executive of the Association of the British Pharmaceutical Industry...business leaders are urging Prime Minister Theresa May to finalize key Brexit decisions, many are already conducting costly preparations for a rocky break with the EU as the U.K. hurtles toward exiting the bloc in March 2019...The industry is concerned about possible delays in evaluating new drugs when the European Medicines Agency, the bloc’s regulator, moves to Amsterdam from London, he said. The agency is bracing for higher-than-anticipated staff departures due to a host of issues, including the effect of local labor laws on short-term contracts, EMA Executive Director Guido Rasi said in an interview...
- Novartis won’t face Swiss criminal probe over payments to Trump lawyer (biopharmadive.com)
The Swiss attorney general's office will not initiate criminal proceedings against Novartis over the $1.2 million the pharma paid a company controlled by President Donald Trump's personal lawyer Michael Cohen..."Following a detailed analysis, the OAG concluded that there was insufficient suspicion to justify opening criminal proceedings," the Office of the Attorney General of Switzerland wrote in an emailed statement...Not only was Cohen unable to deliver the sought-after advice, but recent revelations of the agreement led to accusations Novartis paid money to gain favorable treatment by the U.S. government...the deal was in place while Novartis was negotiating with the Centers for Medicare and Medicaid Services on reimbursement for its CAR-T cancer therapy Kymriah...
- 5 Ways Pharmacists Can Help Prevent Suicide (pharmacytimes.com)How Suicide Quietly Morphed Into a Public Health Crisis (nytimes.com)
With the recent deaths of Spade and Bourdain, health care professionals are also concerned about suicide contagion, a phenomenon in which high-profile suicides influence patients to attempt or committ suicide themselves...pharmacists can play a key role in preventing suicide...pharmacists are ideally situated to assist those in need because of their frequent interactions with patients and access to medical records...However, pharmacists are often unprepared to properly respond to signs of suicide risk, as very few pharmacy schools incorporate suicide prevention courses into their curricula...here are 5 ways pharmacists can help patients who are contemplating suicide...
- Identify at-risk patients.
- Monitor medication use and mental health.
- Collaborate with the health care team.
- Refer to suicide prevention resources.
- Be encouraging and empathetic.
- This Week in Managed Care: June 8, 2018 (ajmc.com)
Kelly Davio, welcome to This Week in Managed Care from the Managed Markets News Network
- After setting aside $200M, Endo settles 1,300 testosterone liability lawsuits (fiercepharma.com)
...Endo International has inked a deal to put 1,300 testosterone drug liability lawsuits to rest...The company disclosed a "master settlement agreement"...to resolve "all known" cases it faces. It will pay into a settlement fund, from which plaintiffs can release their claims...The deal doesn't include an admission of wrongdoing...Late last year, the company added $200 million to its legal reserves to cover costs in testosterone product liability litigation. The company reported in May that it faced about 1,300 testosterone cases. About 900 suits are in a nationwide multidistrict grouping against Endo and several other drugmakers...The plaintiffs alleged that Endo and other drugmakers overmarketed their testosterone drugs and ignored safety risks, leading to serious health complications...The company is among a group of drugmakers facing thousands of lawsuits alleging harm from testosterone replacement therapies. AbbVie faces about 4,600 lawsuits in the multidistrict litigation...while Eli Lilly faces about 500. All told, the multidistrict litigation includes about 7,700 testosterone liability lawsuits. Eli Lilly previously entered a memorandum of understanding about a potential settlement over lawsuits alleging harm from Axiron.
- Medicare Part D drug spending spikes by 77% (healthcarefinancenews.com)
Despite a decrease in the number of prescriptions for brand-name drugs going down, Part D spending and out-of-pocket costs both spiked up from 2011 to 2015, according to a new report from the Office of the Inspector General...The total reimbursement for all brand-name drugs in Part D rose by 77 percent from 2011 to 2015 even though there was a 17-percent drop in prescriptions for those drugs. Even after taking into account manufacturer rebates, reimbursement for Part D brand-name drugs still swelled by 62 percent during that time period,...the number of beneficiaries shouldering at least $2000 in out-of-pocket costs per year nearly doubled across the five-year time span and unit costs for brand-name drugs rose nearly 6 times faster than inflation...for small, rural or struggling hospitals whose razor thin margins are already the source of angst for c-suiters, watching drug spend totals steadily rise could force painful decisions like opting to not modernize EHRs or update equipment...
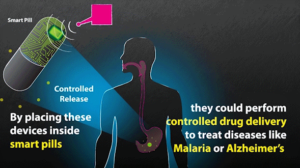
- Wireless system can power devices inside the body (news.mit.edu)
MIT researchers, working with scientists from Brigham and Women’s Hospital, have developed a new way to power and communicate with devices implanted deep within the human body. Such devices could be used to deliver drugs, monitor conditions inside the body, or treat disease by stimulating the brain with electricity or light...The implants are powered by radio frequency waves, which can safely pass through human tissues...in animals, the researchers showed that the waves can power devices located 10 centimeters deep in tissue, from a distance of 1 meter...Even though these tiny implantable devices have no batteries, we can now communicate with them from a distance outside the body. This opens up entirely new types of medical applications...An overarching aspiration is that regulators will provide input to the design and may incorporate framework elements and learnings into regulatory programs. .
- Blockbuster Drugs? How About Doing More With What We’ve Got (bloomberg.com)
When it comes to innovation in health care, it's easy to focus on potential blockbuster drugs that hold the promise of flashy cures with billion-dollar sales potential. But a growing crop of research suggests there are also gains to be made by better using drugs we already have...Medical research as a whole is risky, takes time and can be expensive — so it isn’t surprising that studies involving existing drugs often don't get the sort of funding or attention that potentially curative new treatments do. But at a time of spiraling drug prices, they should be a greater priority for governments and other entities interested in keeping down health-care costs without sacrificing patient care...The problem is funding. Pharmaceutical companies have less of an incentive to put their research dollars in these types of studies because there’s rarely fresh profit involved...
- Gottlieb Proposes Modernization of Drug Review Office (biopharminternational.com)
...FDA Commissioner Scott Gottlieb, MD, announced that the Center for Drug Evaluation and Research would be taking steps to modernize the organization and functions of the Office of New Drugs in order to address scientific and medical advances within the industry. The goal is to make the review process more integrated across science and regulatory expertise. Janet Woodcock, director of CDER, plans on elevating the role of FDA scientists and medical officers and providing these officers with more tools and support “to advance the clinical and regulatory principles that the FDA uses to evaluate new drugs for safety and efficacy.”...Other changes will include the development of guidance documents, giving review staff more time with sponsors, and getting sponsors involved earlier in the development process. Engaging disease specialists, academic researchers, regulatory partners at other agencies, and patient groups is also a goal of CDER.
- ISPE Field-Tests Quality Program (biopharminternational.com)
The International Society of Pharmaceutical Engineering announced...that it is field-testing the design principles of a comprehensive industry-led program of self-evaluation of pharmaceutical quality program with industry colleagues, senior leaders, and regulators. The vision for the program is to evolve the focus from submission of harmonized quality metrics as given in recent FDA guidance documents to establishing a framework for advancing the state of pharmaceutical quality, while at the same time continuing to align with the purpose outlined by FDA...ISPE believes this approach will deliver on the objectives outlined by FDA and will increase the value to industry...