- Exclusive: FDA plans new compounding pharmacy policy, agency head says (reuters.com)
The head of the U.S. Food and Drug Administration said...the agency is working on a new policy that would encourage more compounding pharmacies to register under...the Drug Quality and Security Act, which aimed to bring more compounding pharmacies...under the authority of the FDA rather than state pharmacy boards...The law created a category of “outsourcing facilities” that could register with the FDA, allowing them to sell products in bulk to hospitals and physician practices without prescriptions for individual patients...In exchange, those compounders would have to follow federal manufacturing standards and subject themselves to routine inspections...around 70 firms have registered as outsourcing facilities...compounders that did not register with the FDA would remain under state oversight, and...could only compound drugs based on prescriptions for specific patients...Gottlieb said that in order to encourage more compounders to register, the FDA would release draft guidance in the next two months reflecting its intention to adjust its enforcement priorities based on the size of registered compounders and the riskiness of their products...We’re looking at ways we can provide more of a gradation in our regulatory architecture so we don’t have a one-size-fits-all approach...
- This Week in Managed Care: September 15, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- The FDA just approved the first app for treating substance abuse (cnbc.com)
Federal regulators...approved the first mobile app to help treat substance use disorders...The app, developed by a start-up called Pear Therapeutics, is designed to be prescribed by clinician and used alongside counseling...Pear's technology digitizes a form of talk therapy called cognitive behavioral therapy, or CBT, which focuses on "examining the relationships between thoughts, feelings and behaviors...Pear Therapeutics is part of a burgeoning category of health start-ups known as digital therapeutics. The idea is that software can improve a person's health, without the same cost and side effects of medical treatment...Pear's app has not been approved to treat opioid dependence, but...the company has developed a version of the software that is currently under submission. It's designed to be used alongside opioid replacement therapies.
- Increased use of pot by pregnant women spurs Nevada campaign (reviewjournal.com)
The state of Nevada is preparing a public information campaign to address the increasing use of marijuana by pregnant women and highlight the potential harm the drug can do to a fetus...The public service TV and radio ads, which will begin airing in December, come as research shows that more pregnant women are using pot. A federal study last year found that marijuana use by pregnant women in the U.S. increased from 2.4 percent in 2002 to 3.9 percent in 2014, a 62 percent jump...Studies in states that have legalized recreational pot suggest the rate of use is far higher...In Colorado, where marijuana was legalized in 2014, one Pueblo hospital reported that the number of babies born with the chemical effects of marijuana in their systems doubled in two years. Dr. Larry Wolk, executive director and chief medical officer of the Colorado Department of Public Health and Environment, called the report anecdotal but noted that the state’s most recent official survey found that 6 percent of pregnant women were using marijuana.
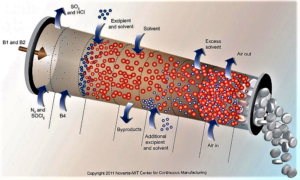
- FDA calls for industry input on continuous manufacturing guidelines (biopharmadive.com)
The Food and Drug Administration plans to develop clearer guidelines around the adoption of continuous manufacturing and has asked industry stakeholders for input on how best to design a regulatory framework...The regulator has advocated companies make the switch from traditional batch production to continuous manufacturing, which to a large extent hasn't been widely adopted by drugmakers despite the potential benefits to speed and reliability...continuous manufacturing can reduce the risk of manufacturing failures and potentially help prevent drug shortages from developing — areas that have been in focus as the agency steps up its oversight of pharmaceutical production facilities...Pharma has been loath to change over from batch production, which has served the industry for decades...Continuous manufacturing techniques — already adopted in many other industries — promise to shorten production times significantly, while reducing the amount of human intervention needed throughout...The FDA hopes providing a "framework of principles" will help drugmakers navigate CM adoption and implementation...
- Week in Review: September 15, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Martin Shkreli’s new jailhouse home is ‘not where you want to be’: Defense lawyer (cnbc.com)
Shkreli is expected to be locked up...for four months...Martin Shkreli's new home in a Brooklyn, New York, federal jail is definitely not the kind of place "where you want to be,"…Metropolitan Detention Center Brooklyn...Shkreli...was thrown into the MDC...after a Brooklyn federal court judge revoked his $5 million release bond...Judge Kiyo Matsumoto ruled that Shkreli represented a danger to the community because of a bizarre $5,000 bounty he offered to Facebook followers who grabbed samples of Hillary Clinton's hair for him...Shkreli, who previously ran two pharmaceutical companies and several hedge funds...A jury convicted him in August of three securities fraud charges...
- Drug industry on tenterhooks as Maryland price-gouging law nears (reuters.com)
...state authorities and patient advocates in Maryland are preparing to enforce the nation’s first law designed to punish drugmaker price-gouging...The state Attorney General’s office said it will field complaints and investigate “unconscionable increases” in essential generic medicines when the closely watched law takes effect Oct. 1...Drugmakers fear the Maryland law will embolden other states and are seeking a court injunction. Both sides made their arguments...before a U.S. District Court judge in Baltimore, who could decide on an injunction in the coming days...But states, struggling to cover rising healthcare costs, are taking up the fight. At least 176 bills on pharmaceutical pricing and payment have been introduced this year in 36 states, according to the National Conference of State Legislatures...Maryland’s law is the most aggressive legislation to be passed so far, and allows the state to levy fines and order a reversal of price increases...
- Challenge of Allergan tribal patent deal in uncharted legal territory (reuters.com)
As generic drug manufacturers are gearing up to argue that a deal Allergan Plc made with a Native American tribe to shield patents from administrative review is a sham, some experts say the generic companies are in uncharted legal territory...Last week, Allergan announced it would transfer the patent rights to its Restasis dry-eye treatment to the Saint Regis Mohawk Tribe, which will license them back to the company in exchange for ongoing payments...Richard Torczon, a lawyer for generic drug company Mylan NV, said the tribe is abusing the defense of sovereign immunity, which he said is intended to shield tribes that get dragged into court without their consent...“The tribe here has not been dragged into this proceeding against its will,” Torczon said during a hearing...before three judges from the patent board. “It has deliberately by its own admission targeted these proceedings for exactly this kind of revenue-generating opportunity,”
- Discrepancy between trial goals, results may mask treatment risks (reuters.com)Association of Trial Registration With Reporting of Primary Outcomes in Protocols and Publications (jamanetwork.com)
Scientists often fail to publicly register plans for clinical trials or to publish the results, and the outcomes they do share may mask instances when new treatments are unsafe or ineffective, a small study suggests...One goal of asking scientists to register clinical trial plans in public research databases is to highlight the main objective of experiments and make it easy to see whether a tested treatment achieved this goal when results are published, the study authors note in JAMA...But more than one-third of the 113 clinical trials researchers examined for the study were never registered. Only 64 of the trials, or 57 percent, were published...One in five trials didn’t define a primary outcome, or clear protocols for determining if the tested treatment had achieved its main goal...Doctors rely on published evidence to guide patient care decisions, while researchers use the published literature to guide which promising areas of inquiry to pursue...Unpublished trials tend to be the ones that found treatments didn’t work or weren’t safe...Published results, meanwhile, tend to highlight successful experiments...