- Genentech lobbyist’s life and death makes pharma waves (fiercepharma.com)
A Genentech lobbyist who generated enormous sales for the company allegedly kept millions in consulting payments for himself and ended his own life after authorities and the company began looking into the charges...During his time lobbying for Roche and its Genentech unit, Evan Morris’ tactics boosted the company's top line by hundreds of millions of dollars, with the company's flu drug Tamiflu and cancer treatment Avastin among the products that benefited...Under investigation for embezzlement in July 2015, Morris committed suicide at a Virginia golf course...Morris’ lavish life, ...featured expensive houses, wines, influential contacts and more, before it came quickly crashing down. An obituary for Morris simply says he “resided in Alexandria, VA.” He was born in 1977 and died on July 9, 2015, the obituary says.
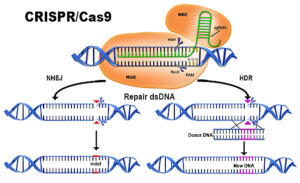
- Broad Institute notches win in CRISPR patent battle (biopharmadive.com)
In a hotly anticipated judgment, the U.S. patent office...ruled that key patents held by the Broad Institute of MIT and Harvard on the gene-editing technology CRISPR-Cas9 do not overlap with competing claims from the University of California...UC may now receive a patent covering broader claims on the revolutionary tech...the Patent Trial and Appeal Board ruled the claims of UC and the Broad to be patentably distinct subject matter, leading the three judges to declare a judgment of non-interference...Put into layman's terms, the claims of the UC and the Broad are distinct enough so as to be separately patentable and therefore don't overlap with each other. This effectively supports the Broad's existing patents, while sending the UC's pending patent application back to the initial patent examiner...UC with a patent on use of CRISPR in all cells and the Broad with a patent for use of CRISPR specifically in eukaryotic cells...If that were to happen, biotechs operating in the space could end up having to license the tech from both UC and the Broad.
- Pharma Lobby May Boot Company That Introduced High-Priced Drug (bloomberg.com)
The Washington lobbying group for the pharmaceutical industry may kick out a drugmaker that introduced a high-priced version of an older medicine that had been available much more cheaply...Pharmaceutical Research and Manufacturers of America...is reviewing its membership criteria after Marathon Pharmaceuticals LLC...said it would introduce a drug for a rare, deadly muscle disease at the cost of $89,000 a year. A version of the drug had been available for about $1,000 a year overseas and many U.S. patients were allowed to import it...Marathon’s...recent actions are not consistent with the mission of our organization...The leadership of the PhRMA board of directors has begun a comprehensive review of our membership criteria to ensure we are focused on representing research-based biopharmaceutical companies that take significant risks to bring new treatments and cures to patients…
- Aetna, Humana terminate $37 billion merger (chaindrugreview.com)
After being blocked by a federal court, Aetna Inc. and Humana Inc. have decided to terminate their $37 billion merger deal...The health insurance giants said...that, with the move, Aetna will pay Humana a $1 billion breakup fee...In January...the...District Court for the District of Columbia ruled in favor of the Department of Justice’s request to enjoin the Aetna-Humana merger. The government claimed that the combination of the two health insurers would lessen competition, harming seniors who buy private Medicare coverage and some consumers who buy health insurance on public exchanges...The consolidation wave hitting the health insurance sector has been seen as a byproduct of the Affordable Care Act. The mega-deals would provide fast access to the millions of people who have gained health coverage under the ACA and would be a vehicle for capturing Medicaid business, which has expanded dramatically under the health reform law...the climate for mergers and acquisitions in the health care sector has become uncertain. Besides the DOJ’s opposition to the mega-mergers, the Trump administration and Republican leaders in Congress have begun efforts to dismantle the ACA. That has cast a cloud on the health insurance marketplace, since it remains unclear what would replace the ACA and how consumers who have obtained coverage via the exchanges would continue to receive benefits.
- Pharma industry shuns Trump push for radical shift at FDA (reuters.com)
President Donald Trump's vow to roll back government regulations at least 75 percent is causing anxiety for some pharmaceutical executives that a less robust Food and Drug Administration would make it harder to secure insurance coverage for pricey new medicines...Industry trade group Biotechnology Innovation Organization told Reuters...lobbyists urged the administration not to name a new commissioner of the Food and Drug Administration who would act rashly to speed up the agency’s approval of new medicines...the FDA is already adopting new drug development models and warned that a looser review process would put patients at risk...People often argue that the FDA is too restrictive...We have the sense that the balance is pretty right...you have to have a well-characterized risk/benefit profile."...While most sectors welcome less oversight, drugmakers say a robust review process is critical in convincing physicians and insurers that a pricey new medicine has value...Otherwise, the time and money it takes to get a new drug to market - estimates run as high as $2.6 billion - would be lost if insurers are not willing to pay for the product... payers are looking for evidence of value...
- This Week in Managed Care: February 17, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Pharmacy Week in Review: February 17, 2017 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharma lobby launches crusade against Cuomo’s ‘flawed’ drug pricing plan in NY (fiercepharma.com)
Pharma has heard enough of New York Governor Andrew Cuomo’s ideas for lowering drug prices. Just a month after the governor unveiled his plan, the industry’s trade group is rolling out a counteroffensive urging state officials to “Prescribe Real Solutions”...Gov. Cuomo’s plan would create a board to rule on “fair prices” for certain expensive drugs. Then, the state would mandate that Medicaid refuse to pay more than that for those meds...Cuomo's proposal also factors into private market sales. If a pharma company sold into New York at a higher price than the board’s "fair price," the state would collect a surcharge...PhRMA is having none of that. The group’s Prescribe Real Solutions website asks the public to “demand real solutions” from their lawmakers...PhRMA’s deputy vice president for state advocacy in New York, maintains that Cuomo's proposal "does nothing to improve patient access to medicines, bring life-saving drugs to the market or create jobs" in the state..."This is not the time for unworkable proposals that would create new bureaucracies without actually addressing any of the factors that impact the prices paid by patients,"...
- Pfizer, Flynn appeal against UK regulator’s £89.4m fine for epilepsy drug price hike (manufacturing.pharmaceutical-business-review.com)
Pfizer and Flynn Pharma have appealed against the £89.4m ($111m) fine imposed by the UK Competition and Markets Authority (CMA) for charging excessive and unfair prices for an anti-epilepsy drug...Pfizer has been fined £84.2m($104m) and Flynn Pharma £5.2m ($6.4) after they increased prices by as much as 2,600% in September 2012...The companies have separately filed appeals in the Competition Appeal Tribunal...Both Pfizer and Flynn accused the UK watchdog of ignoring market realities when it ruled that the price Flynn paid to acquire the anti-epilepsy drug from Pfizer and the price it charged from the National Health Service (NHS) were both excessive...The CMA had stated that owing to the unreasonable price increase of the critical drug, the NHS’ annual expenditure to buy the phenytoin sodium capsules from Flynn had shot up to around £50m ($62m) in 2013 from the 2012 figure of £2m ($2.4).
- Express Scripts Reveals What Really Drives Drug Spending—And Why the Government May Do No Better (drugchannels.net)
Last week, Express Scripts released its 2016 Drug Trend Report for the commercial market…For 2016, drug trend—the year-over-year change in spending—was a mere 3.8% for Express Scripts’ commercial plan sponsor customers. The Express Scripts data also highlight how drug prices were only part of the reason behind moderating drug spend. Contrary to the public rhetoric, increased utilization was a bigger influence than higher prices for both specialty and traditional drugs...The new Express Scripts report deconstructs year-over-year changes in drug spending into its two primary components:
- Unit costs—the payer’s cost per unit of therapy. Unit costs vary with: 1) the rate of inflation in brand-name drug prices, 2) shifts to different drug options within a therapeutic class, 3) a shift in mix of therapeutic classes utilized by beneficiaries, and 4) the substitution of generic drugs for brand-name drugs. Note that for the second year in a row, the Express Scripts unit cost data account for the value rebates shared with payers.
- Utilization—the total quantity of drugs obtained by a payer’s beneficiaries. Utilization varies with: 1) the number of people on drug therapy, 2) the degree to which people adhere to their drug therapy, and 3) the average number of days of treatment.