- Burden of pricey hepatitis C drugs heaviest in eastern Europe (reuters.com)Prices, Costs, and Affordability of New Medicines for Hepatitis C in 30 Countries: An Economic Analysis (journals.plos.org)
New hepatitis C drugs that can cure the liver-destroying disease are revolutionizing treatment but their high price is a huge burden in some countries, particularly in central and eastern Europe, according to a new study...An analysis of prices for two Gilead drugs by World Health Organization...found that while U.S. prices were higher in dollar terms, parts of Europe paid considerably more in "purchasing-power parity" terms...The PPP-adjusted price in Poland of a treatment course with Gilead's Harvoni was $118,754, against $72,765 in the United States...and it would cost 190.5 percent of the country's total drugs budget to treat all Polish patients...Gilead says the price of its treatments reflect their clinical, economic and public health value, arguing they are cost-effective over the long term.
- Most scientists believe there is a ‘crisis’ reproducing experiments (statnews.com)1,500 scientists lift the lid on reproducibility (nature.com)
Placing trust in science can be easier when findings are confirmed, but a new survey finds that most scientists believe there is a "crisis" in reproducibility...Specifically, 52 percent reported that replicating results is a "significant" problem and another 38 percent believe a "slight crisis" exists. More than 70 percent of researchers have tried and failed to reproduce another scientist’s experiments, according to Nature, which canvassed 1,576 researchers. And more than half of the respondents reported that they failed to reproduce their own experiments...Yet one-third believe that failing to reproduce results means that a study is probably just incorrect, and most of those asked say that they continue to trust published findings. Moreover, 73 percent think that at least half of the papers in their own fields can be believed...Merck executive recently suggested that drug makers should be entitled to get their money back for potential treatments licensed from universities if the company is unable to reproduce the results in subsequent experiments...the pharmaceutical industry has been under pressure to release trial results in order to verify claims about their medicines. The issue accelerated in the wake of several scandals about undisclosed side effects...
- Why taking morphine, oxycodone can sometimes make pain worse (sciencemag.org)
There’s an unfortunate irony for people who rely on morphine, oxycodone, and other opioid painkillers: The drug that’s supposed to offer you relief can actually make you more sensitive to pain over time. That effect, known as hyperalgesia, could render these medications gradually less effective for chronic pain, leading people to rely on higher and higher doses. A new study...the first to look at the interaction between opioids and nerve injury for months after the pain-killing treatment was stopped—paints an especially grim picture. An opioid sets off a chain of immune signals in the spinal cord that amplifies pain rather than dulling it, even after the drug leaves the body...Yet drugs already under development might be able to reverse the effect...spinal cord...microglia—sentinels of the nervous system that scout for infection...release inflammatory signaling molecules into the spinal cord, which activate neurons that shoot pain signals up to the brain...Researchers are...exploring drugs that interrupt this pathway to treat pain or improve the performance of opioids. A clinical trial recently launched at Yale University, for example, will test whether an antibiotic that inhibits glial cells prevents the inflammatory effects of opioids.
- Most Americans would avoid clinical trials due to worries over safety and costs (statnews.com)
Most Americans would not enroll in clinical trials over concerns they would experience side effects, encounter higher costs, or receive a placebo instead of an actual medicine, according to a new survey...only 35 percent say they were likely to participate in a study and, overall, just 40 percent have a positive view of clinical trials, according to the survey...conducted by the Memorial Sloan Kettering Cancer Center...The results underscore the difficulties that confront policy makers, pharmaceutical companies, and health care providers as they seek to develop and deliver new treatments. And if this sort of reluctance to participate in trials continues, there is concern there will be a research and discovery "crisis,"...clinical trials are the engine that drives innovation...This is a critical element in the process...We have so many new agents and opportunities to improve outcomes. But if we don’t enroll people in clinical trials, we will not realize the benefits...
- Heavyweight champion of the world (pharmatimes.com)
For more than 15 years NICE has punched above its weight internationally but as it comes under attack for its methodology, will its international clout suffer?...The bad headlines are back; on 20 May, the National Institute for Health and Care Excellence...came under fire from the media for its decision to bar Roche's Perjeta (pertuzumab) for breast cancer from the NHS...While patient groups and charities are worried about the effect on cancer patients in England, who may be denied treatment, there may also be a knock-on effect in other countries, which still look to NICE for guidance over their own reimbursement decisions...NICE International...advises countries from Ghana to Kazakhstan on HTA (health technology assessment) methodology and implementation...Yet, its international spread has come hand-in-hand with the growing disillusionment about NICE's decisions...An IMS Institute study in 2013 compared the reimbursement of cancer drugs in five countries that used cost per QALY (NICE's quality-adjusted life years) methodologies, including the UK, with five using broader methodologies, including Germany and US. It found that the cost per QALY countries reimbursed fewer cancer drugs, had slower access to those they did adopt, and generally performed poorer in terms of cancer outcomes. Moreover, it was not clear that they saved much money.
- India’s Sun Pharma gets U.S. subpoena over generic drugs pricing (finance.yahoo.com)
U.S. Department of Justice has subpoenaed India's largest drugmaker Sun Pharmaceutical Industries Ltd seeking information about the pricing and marketing of the generic drugs it sells in the United States...The DoJ's antitrust division has also asked Sun Pharma's U.S. unit for documents related to employee and corporate records and communications with competitors...They specifically cited doxycycline hyclate 100 milligram, an antibiotic for which the price doubled in the year through June 2014...Other generic drugmakers including India's Dr Reddy's Laboratories Ltd and U.S. firm Allergan Plc also received subpoenas from regulators seeking similar information last year, but they did not disclose the names of the products involved...
- Roche and Novartis face off in biosimilar drug battle (reuters.com)
Switzerland's biggest drugmakers are clashing over cheaper copies of pricey biotech drugs - one reason why Novartis is considering selling its $14 billion stake in...Roche...With a copycat of Roche's blood cancer drug Rituxan pending European approval, Novartis aims to muscle in on a share of sales that last year hit 7 billion Swiss francs ($7.1 billion)...But Roche is fighting back with a new medicine, Gazyva, which it contends is better than Rituxan...Beyond its own new drug portfolio, Novartis has a big side bet that cheaper "biosimilars" from its Sandoz generics unit can grab rivals' profits, while Roche has limited its focus to new drugs to counter such incursions.
- Rite Aid improves distribution in Southeast with new DC (drugstorenews.com)
Rite Aid announced...the grand opening of its new distribution center in Spartanburg, S.C....first in 16 years," stated John Standley, Rite Aid chairman and CEO. "Featuring highly efficient and advanced technologies, this facility will play a crucial role in our company’s supply chain, supporting more than 1,000 Rite Aid stores across the southeastern United States, and help us deliver a superior customer experience."...approximately 600 new jobs...will have a tremendous impact on the entire state and is a testament to the competitive business environment...The 900,000 square foot distribution center sits on 97 acres...Some of the features of the new facility include:
- High efficiency LED and T5 fluorescent light fixtures and occupancy sensor controlled warehouse lighting;
- An on-site truck maintenance facility, including a fueling station, trailer weigh scale and wash area;
- High-speed automated palletizers, ergonomically designed manual palletizing stations and automatic label applicators;
- State-of-the-art warehouse and labor management system;
- Voice pick technology; and
- 60,000 square feet of office space, including an associate cafeteria, fitness center and other amenities.
- Washington state told to lift restrictions on hepatitis C medicines (statnews.com)
The Washington state Medicaid program has been ordered to lift restrictions on coverage of pricey hepatitis C treatments, according to a preliminary injunction issued...by a federal judge...The ruling came in response to a lawsuit filed by state residents who claim the drugs are "medically necessary," and that the decision by the Washington State Health Care Authority to provide coverage to only the sickest patients had caused them harm...In a strongly worded...opinion, United States District Court Judge John Coughenour agreed with their argument. He wrote that the facts "clearly favor" their contention that state policy violates federal law. In his view, the evidence "establishes that there is a consensus among medical experts and providers that the life-saving [drugs] are medically necessary" for all hepatitis C patients...This is the first time a federal court has declared that widespread restrictions across a state Medicaid program is illegal for hepatitis C medicines...The opinion is only the latest instance in which a public or private insurer has been forced to reconsider its coverage policies toward hepatitis C medicines, which helped trigger a wave of controversy over the cost of prescription drugs...
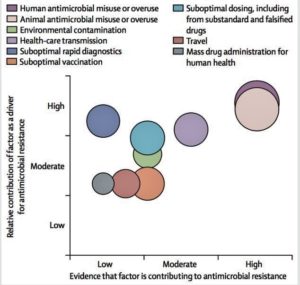
- New incentives needed to develop antibiotics to fight superbugs (reuters.com)G7 told to act on antibiotics as dreaded superbug hits U.S. (reuters.com)Scientists have just found a “nightmare superbug” in the US (vox.com)
Drugmakers are renewing efforts to develop medicines to fight emerging antibiotic-resistant bacteria, but creating new classes of drugs on the scale needed is unlikely to happen without new financial incentives to make the effort worth the investment...American military researchers...announced the first U.S. case of a patient with an infection found to be resistant to the antibiotic colistin, the drug often held in reserve for when all else fails...That put a spotlight on the urgent need for new medicines that can combat what health officials have called "nightmare bacteria."...Drugmakers...acknowledged that in the absence of a new way of compensating them, it simply does not make economic sense to pour serious resources into work on new antibiotics...80 drugmakers and diagnostics companies...signed a declaration calling for cooperation among governments and companies to create incentives to revitalize research and development of new antibiotics...It proposed a new business model in which profit would not be linked to higher sales...