- Those PBM moves pharma loves to hate? They work, CVS says–so get used to them (fiercepharma.com)
Drugmakers may not be too keen on the formulary management techniques and exclusive deals pharmacy benefits managers have been using to combat rising drug prices. But new numbers from a PBM giant suggest they work--and that pharma better get used to them...CVS Health announced that its prescription drug trend--a metric for growth in prescription spending--had dropped to 5% in 2015, down from an all-time high of 11.8% in 2014. And with many of the usual drivers staying consistent over both periods--including brands and price inflation among generic and specialty drugs—the company is chalking up the drop as proof its tactics are doing their job...CVS' "proactive pharmacy management strategies were successful in mitigating the impact of rising drug costs in today's highly volatile prescription drug market,"..."[W]e work to help patients avoid unnecessarily expensive medications as part of our focus on making prescriptions affordable."..CVS and its main rival, Express Scripts, have both been actively pruning their preferred formularies over the past few years…Unsurprisingly, pharma hasn't taken too kindly to the moves, but some companies have been more accepting of the current climate than others...GlaxoSmithKline and Novartis, to name a couple, have been working to avoid payers' wrath...
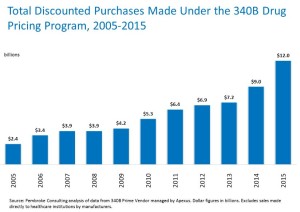
- 340B Purchases Hit $12 Billion in 2015—and Almost Half of the Hospital Market (drugchannels.net)
According to new data...discounted purchases made under the 340B Drug Pricing Program hit $12 billion in 2015. That’s a whopping 67% higher than the 2013 figure. I estimate that the undiscounted value of these purchases exceeds $17 billion...Most 340B purchases are made by hospitals. My...number-crunching below reveals that hospitals now receive 340B discounts on more than 44% of their drug purchases...two years ago, the 340B program is taking over the hospital market...How much of this money goes to uninsured and needy patients? No one knows, and the hospitals aren’t saying much...340B purchases have been growing much, much more quickly than have hospitals’ total drug purchases. From 2005 to 2015, total hospital drug purchases grew by 31%, compared with the 400%+ growth in the total 340B purchases...340B has infiltrated almost half of the hospital market...The 340B program is highly controversial, partly because the 340B legislation does not specify or restrict how covered entities should utilize funds generated by the program. Hence, it’s troubling to see that uncompensated care as a percentage of hospitals’ total expenses has remained at about 6% for many years, despite booming 340B purchases...Defenders of the 340B status quo argue that covered entities should be able to use discounts to reduce their cost of operations, without any transparency or accountability...The Office of Inspector General has documented how 340B-eligible hospital outpatient departments earn tremendous profits from the Medicare Part B program. Gross profit margins are about 60% compared with 3% to 4% for a non-340B outpatient program...
- Management Tools for Controlling Specialty Drug Costs (specialtypharmacytimes.com)
The increasing utilization and costs of specialty drugs will have a substantial impact on overall health care costs during the next decade...Pharmacy benefit manager tools that have successfully controlled costs in the traditional small-molecule drug categories will be critical to manage the increasing costs and requirements of the specialty drug category…
Financial Impact of Specialty Drugs: Current and Projected...by 2020, 9 of the 10 best-selling drugs (by revenue) will be specialty drugs...spending could reach $400 billion, or 9.1% of national health spending...Specialty benefit design and management tools employed by PBMs will also play a significant role in controlling specialty drug spending, as well as ensuring optimal patient care and support.
Specialty Benefit Design and Management...Specialty drugs frequently have unique shipping and storage needs, as a result specialty pharmacies are better equipped to procure, store, and dispense these treatments than traditional retail pharmacies. Furthermore, pharmacists and personnel at specialty pharmacies provide patient education and clinical support beyond the capabilities of a retail pharmacy...The tools in the PBM arsenal are critical to ensure appropriate care for patients needing specialty drugs while managing the often extraordinary costs.
Comprehensive management approaches that monitor and balance patient care outcomes and costs will help PBMs ensure that new, innovative medications are readily available and affordable to the patients who need them most.
- Senate confirms Dr. Robert Califf to lead FDA (reuters.com) New FDA head Robert Califf vows to use ‘bully pulpit,’ better explain agency decisions (washingtonpost.com)
The Senate voted overwhelmingly...to confirm Dr. Robert Califf as head of the Food and Drug Administration, an agency that regulates everything from food and drugs to tobacco, cosmetics and dietary supplements...Califf...a well-regarded cardiologist and researcher, takes the helm at the FDA when lawmakers are pressuring it to speed the approval process for drugs and medical devices and to finalize a proposed rule giving it authority to regulate e-cigarettes...He said one of his first priorities is to strengthen the workforce by reaching out to academic and other centers to attract new talent...Another priority...is improving surveillance systems to monitor for safety...We're not proposing to do away with the adverse event reporting system that currently exists...but we are acutely aware that it is not enough...Tools to monitor the safety of medical devices also need to be modernized...and though it will not happen overnight...we have to do the hard work of making it happen...
- How should FDA revamp its off-label marketing rules? An expert panel backs big changes (fiercepharma.com)
After a series of court decisions put its regulatory approach in question, the FDA promised new guidance on off-label drug promotions. That was more than a year ago. Now, a group of health policy wonks and legal experts has come out with some suggestions...The recommendations from the Duke-Margolis Center for Health Policy include one big change that might simplify the free-speech debate over off-label marketing. The agency could revamp its approach to official labeling to include clinical data on off-label uses, graded for evidence quality, and even post-marketing data generated through the agency's own Sentinel surveillance system…Changing official FDA labeling...the Duke-Margolis Center suggests using pilot programs...One might use the Sentinel system. Another might allow provisional off-label use claims based on evidence that's not enough for a new indication, contingent on Sentinel keeping watch on the new use. Overall, the idea would be to develop a tiered labeling system, with the strongest supporting data on the highest tier, and lesser quality evidence farther down...more radical suggestion? Setting up a third-party data-review organization with the FDA's involvement...[That] would assess new off-label promotional material and give it a quality-control score based on the quality of evidence backing it up…The FDA clearly wants to roll out its own guidance, rather than letting the courts decide. In December, the agency settled an off-label marketing dust-up with Pacira over its Exparel marketing, and it's on the verge of a deal in Amarin's off-label lawsuit involving its cardiovascular med Vascepa.
- Challenges Independent Pharmacists Face with Customer Retention (pharmacytimes.com)
Dan Benamoz, RPh, President and CEO of Pharmacy Development Services, discusses the challenges independent pharmacists face with respect to customer retention efforts
- Should Pharmacists be called Doctors? – PLR 4 (pharmacyliferadio.com)
Today we touch on a TOUCHY subject: should pharmacists call themselves doctors?...Lots of people have opinions on this, and there is a lack of facts to cut thru the fog of confusion...(podcast 20:36) Today we:
- review the major reasons why pharmacists SHOULD call themselves doctors
- review the reasons why pharmacists SHOULDN’T call themselves doctors
- what we think
- and what you should do to figure out for yourself
- The biosimilar ploy that could save billions in health care costs (statnews.com)
As drug makers race to develop cheaper versions of complicated biologic medicines, some companies are pursuing a tactic that could prove a win for themselves, patients, and the health care system as a whole...They are running studies designed to convince doctors and insurers that patients can be easily switched from expensive biologics...to so-called biosimilars, which are almost identical variants. Their goal is to encourage these kinds of switches without waiting for the Food and Drug Administration to decide whether a particular biosimilar has the exact same clinical benefit as its expensive, brand-name counterpart...Standard FDA approval is good enough for physicians to write prescriptions, but only an "interchangeable" designation will enable pharmacists to substitute a biosimilar for a brand-name biologic without contacting doctors for permission first...more and more companies are eyeing switching studies as a shortcut to expanded market share...drug makers are pursuing this workaround that, as an upshot, could bring lower-cost medicines to more people...
- What Pharmacy Students Need to Know about MTM (pharmacytimes.com)
Medication therapy management has become an important buzzword in the world of pharmacy. Theoretically, MTM serves as a route of communication between pharmacists and patients, allowing patients to gain insight and autonomy over the medications they are prescribed, as well as the opportunity to take an active role in their own health care. Students currently enrolled in colleges of pharmacy across the country are being taught how to conduct an MTM session with a patient and how such a session will contribute to improved health outcomes...but how effective is MTM in a real-life retail setting?...As pharmacy students...we conducted interviews with pharmacists in a variety of community settings to explore just how prevalent the practice is in the average workday...we aimed to address the perceived barriers, successes, goals, and areas for improvement related to MTM. Below are the interview questions along with the most prevalent findings.
- What has been successful in your MTM procedures, outcomes, and process?
- How has MTM led to improved patient compliance, better pharmacist–patient relationships, and improved disease state management?
- What are the biggest barriers to providing MTM services?
- What can be changed to improve MTM to make it more successful?
- How can pharmacy students get involved with MTM?
- What is the goal of MTM?
- ASHP Expresses Major Concerns with Proposed Revisions to Chapter <797> Standards, Offers Recommendations (ashp.org)
A number of proposed changes to the U.S. Pharmacopeial Convention General Chapter <797> Pharmaceutical Compounding — Sterile Preparations standards are incompatible with the medication-use process in patient care environments, ASHP stated in comments submitted to a USP expert panel earlier this month...ASHP points out that many of the new requirements are more appropriate for making drugs from bulk chemicals in compounding pharmacies or outsourcing facilities than for preparing medications for administration in patient care settings. The letter urges the expert panel to revise the proposals to better meet the urgent and unpredictable demands of the typical acute patient care setting...ASHP’s letter encourages USP to reorganize the standards into separate sections covering basic and advanced compounding as well as the preparation of special compounded sterile products such as radiopharmaceuticals and allergenic extracts.