- Pharmacist Discusses Drug Abuse in HBO Documentary (pharmacytimes.com)
A Massachusetts pharmacist is speaking out about prescription drug abuse and heroin addiction in a new HBO documentary...Lauren Heroux-Camirand, RPh, conducted 2 interviews with the filmmakers of "Heroin: Cape Cod USA" at Country Square Pharmacy in Attleboro, Massachusetts...she thought it was important to talk openly about illicit drug use... “We are here, as pharmacists and humans, to be compassionate and help in any way we can,”...“That is why I agreed to participate and speak up.”...In her interviews with the filmmakers, Heroux-Camirand discussed both facts about drug abuse and her experiences seeing people struggle with addiction...The movie will premiere in Massachusetts before debuting on HBO on December 28, 2015
- Prevention Strategies Pharmacists Can Use to Reduce Errors (pharmacytimes.com)
Natasha Nicol, PharmD, FASHP, director of Global Patient Safety Affairs at Cardinal Health, provides prevention strategies to reduce medication errors.
- Survey: Employers taking action to cut specialty drug coverage (drugstorenews.com)
Employers are taking significant action to reduce their exposure to rising costs associated with specialty medicines...Although pharmacy represents approximately 20% of employer-sponsored medical benefits costs, it is increasing at a rate that accounts for roughly half of medical cost inflation and should be a top priority for employers...The price, utilization and delivery of specialty prescription drugs, many of which require special handling or delivery, are a top pain point for employers...employers are beginning to consider new aggressive approaches... employers have added new coverage and utilization restrictions for specialty pharmacy, such as requiring prior authorization or limiting quantities based on clinical evidence... employers plan to exclude compounds from their benefit coverage...the compounding process results in higher cost and their use may not be FDA-approved in compound form, health insurers increasingly will not cover them...
- Marsh Drugs extends free diabetes medicine program through 2016 (drugstorenews.com)
Marsh Drugs last week extended their Diabetes Care Program throughout 2016...Marsh Diabetes Care supplies seven different diabetes medications for free to any Marsh Drugs patients in Indiana...There is no limit on the number of prescriptions that can be filled and the program covers new prescriptions and refills...The free diabetes medications include:
Glipizide - Glipizide ER - Metformin - Metformin ER - Glimepiride - Glyburide - Glyburide Micronized
As part of the program, Marsh Drugs also provides a free 100-count box of universal lancets with a prescription.
- Under pressure, CDC delays release of opioid prescribing guidelines (statnews.com)Draft CDC Guideline for Prescribing Opioids for Chronic Pain, 2016: Summary of Constituent Comments and CDC Response (freepdfhosting.com)Painkiller politics: Effort to curb prescribing under fire (hosted.ap.org)
Under mounting criticism, the Centers for Disease Control and Prevention has delayed plans to...release controversial opioid prescribing guidelines for primary care physicians. Instead, the guidelines will now be issued sometime later in the year...The delay was first signaled yesterday when the CDC unexpectedly disclosed that public comments on its proposal would be accepted through Jan. 13. However, the agency will also tap the National Center for Injury Prevention and Control’s Board of Scientific Counselors, a federal advisory committee, to review the guidelines and public comments...The postponement follows complaints from representatives of other federal agencies and consumer advocacy groups that the CDC guidelines were based on weak evidence and would unfairly restrict some patients from obtaining needed pain relief. The agency was also chastised for using a “secretive” process to formulate the guidelines, which some critics argued had violated federal law...the CDC delay apparently reflects a desire to comply with the Federal Advisory Committee Act, which governs how expert groups are formed to provide official advice.
- The testing of new drugs is on the rise. Why that worries some people. (washingtonpost.com)
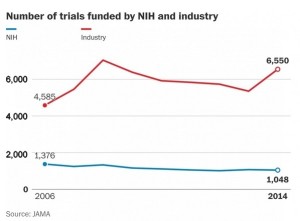
As the budget for the National Institutes of Health, the nation's largest funder of biomedical research, has flatlined over the past decade, the number of clinical trials funded by the federal agency has dramatically declined, a new study has found. At the same time, the number of trials funded by the pharmaceutical industry has exploded...Meanwhile, the number of NIH-sponsored trials fell from 1,376 registered on a website in 2006 to 1,048 by 2014, according to the study, which was published in the Journal of the American Medical Association. Industry-sponsored trials grew by about 2,000 over the same period, reaching 6,550 registered in 2014...More trials is generally a good thing...because it means that more new drugs, devices or other treatments are being tested. But the decline in federally funded trials concerns public health researchers, because federally funded trials are independent, often intended to compare drugs or other treatments instead of simply to show that a product works and should gain federal approval…there are many areas of deserving research...there may need to be a formal discussion of how research dollars are spent. A reasonable way to begin...is to look at the burden of a particular disease and allotting research dollars to those that have the greatest effect on mortality and disability...
- Nonprofit vows to lower generic drug costs (bostonglobe.com)
...in this Central Massachusetts town (Blackstone Valley), a small team of drug industry veterans has launched a startup (Drew Quality Group) to counter...price gougers by making affordable generic medicines to treat critical diseases. And they’re doing it as a nonprofit...We want to create a competitor to stabilize the prices...When you end up with a single-source manufacturer, they can charge any price they want...typically have only a single producer, are in short supply either because their manufacturer is grappling with safety problems or there is no domestic supplier and an overseas producer no longer finds it profitable to make drugs to treat small numbers of patients...By operating as a nonprofit, Drew Quality won’t have to pay the same taxes a drug company does. Nor will it have to focus on cost-cutting by sending work overseas...The group wants to employ people close to home and make sure patients get access to therapies they need without being squeezed financially...And while it plans to underprice companies that have boosted prices substantially, Drew Quality will still charge enough to pay its employees and plow some money back into the business...the cost of its generic drugs will be closer to the discounted prices that were charged before companies pushed them higher.
- NOAC Dosing for Obese Patients vs. Non-Obese Patients (pharmacytimes.com)
Katie Greenlee, PharmD, BCPS-AQ Cardiology, of the Cleveland Clinic discusses how NOAC (novel oral anticoagulants) dosing for obese patients differs from NOAC dosing for non-obese patients.
- FDA launches Web-based precision medicine platform for next-generation sequencing (healthcareitnews.com)FDA Launches precisionFDA to Harness the Power of Scientific Collaboration (blogs.fda.gov)
Food and Drug Administration...launched the beta version of precisionFDA, its new collaborative platform for the exploration of next-generation gene sequencing...First announced in August, the platform features more than 20 public and private sector participants…Next-generation sequencing enables researchers to compile a vast amount of data on a person's exact order or sequence of DNA...scientists can look for meaningful differences in DNA that can be used to suggest a person's risk of disease, possible response to treatment and assess their current state of health. Ultimately, what we learn about these differences could be used to design a treatment tailored to a specific individual...The hope is to grow this community and improve the usability of precisionFDA in the coming months and years...One way we'll achieve that is by placing the code for the precisionFDA portal on the world's largest open source software repository, GitHub, so the community can further enhance precisionFDA's features...
- It’s official: FDA shoots past 2014’s new drugs record with Roche lung cancer med nod (biopharmadive.com)
FDA last Friday approved Alecensa (alectinib) for the treatment of ALK-positive non-small cell lung cancer for patients with the disease refractory to therapy with Xalkori (crizotinib). The approval comes well before the drug's March 2016 PDUFA date...This marks the agency's 42nd newly approved medication this year, topping a banner 2014 that saw 41 new drug approvals...there will be a continuing flurry of drug approvals over the next four years (225, to be exact, and most of them in cancer-related therapeutic spaces).