- Pharmacy Week in Review: November 10, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- FDA commissioner warns drug companies of ‘disruptive’ regulations to fight opioid epidemic (cnbc.com)
The Food and Drug Administration is likely to take new actions on opioids that may be "disruptive" and "uncomfortable" to drugmakers, the agency's commissioner (Scott Gottlieb) said...In addition to seeking to treat opioid-addicted patients with alternative medications that don't produce a high, the FDA says it will look at ways to reduce exposure to the drug. That includes new ways of packaging and distribution..."For example, it's possible that a defined, short-term supply of medication could be packaged in a manner that limits the number of pills dispensed,"..."We're at a point in this crisis that we're going to have to think of ideas and taking actions that are going to be more disruptive and are going to be uncomfortable to some parties," Gottlieb told "Squawk Box." "But we have to take more vigorous action to get ahead of this."...Gottlieb said the agency is having discussions with drug companies about the new packaging solutions..."Something like this could move potentially quickly," he said. "We're invested in taking a hard look at this and seeing what the opportunities are."...
- This Week in Managed Care: November 3, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- FDA approves eight European regulators to inspect drug factories for U.S. (reuters.com)
The U.S. Food and Drug Administration said...it will allow regulators in eight European countries to determine whether drug manufacturing facilities there meet FDA requirements, freeing up American inspectors to spend more time in higher-risk countries like India and China...The FDA said it will begin relying on inspection data from the regulators in Austria, Croatia, France, Italy, Malta, Spain, Sweden and the United Kingdom as of Nov. 1...in some situations the United States could still do inspections in those countries...The move is the first step by the FDA to implement an agreement the United States and the EU finalized in March. The European Commission already determined in June that the FDA could carry out inspections for it in the United States...FDA Commissioner Scott Gottlieb said in a statement that partnering with European regulators would allow the agencies to get “the greatest bang for our collective inspectional buck.”
- U.S. states allege broad generic drug price-fixing collusion (reuters.com)
A large group of U.S. states accused key players in the generic drug industry of a broad price-fixing conspiracy, moving...to widen an earlier lawsuit to add many more drugmakers and medicines in an action that sent some company shares tumbling...The lawsuit, brought by the attorneys general of 45 states and the District of Columbia, accused 18 companies and subsidiaries and named 15 medicines. It also targeted two individual executives: Rajiv Malik, president and executive director of Mylan NV, and Satish Mehta, CEO and managing director of India’s Emcure Pharmaceuticals...The states said the drugmakers and executives divided customers for their drugs among themselves, agreeing that each company would have a certain percentage of the market. The companies sometimes agreed on price increases in advance...“It is our belief that price-fixing is systematic, it is pervasive, and that a culture of collusion exists in the industry,” Connecticut Attorney General George Jepsen, who is leading the case...The...Justice Department is conducting a parallel criminal investigation. On Friday, the department asked the Pennsylvania court presiding over the lawsuit to put the lawsuit’s discovery process on hold, saying it could interfere with the criminal probe
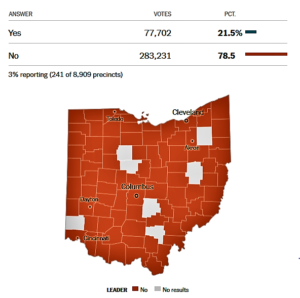
- Ohio Ballot Issue 2: Cap State Agency Drug Costs (nytimes.com)Handing pharma a win, Ohio voters overwhelmingly reject drug pricing measure (fiercepharma.com)
An initiative on Tuesday’s ballot in Ohio is aimed at reducing the cost of prescription drugs in the state. The measure would cap the price of prescription drugs purchased by the state government, including Medicaid...The measure drew strong resistance from drug makers, which spent more than $49 million to try to kill it, and the industry is not accustomed to losing political fights. Last year, it successfully killed a measure in California that was similar to the one in Ohio, but only after spending more than $100 million to do so.
- Pharmacy Week in Review: November 2, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Brexit with no deal risks chaos in drug supply, report warns (reuters.com)‘No deal’ Brexit would harm NHS and its patients, Nuffield Trust warns (nuffieldtrust.org.uk)
Crashing out of the European Union without a deal would cause major problems for Britain’s health service and risk “chaotic disruption” to medicine supplies...The Nuffield Trust, an independent health charity, also warned that Brexit without a deal on future relations with the EU would lead to worsening staff shortages in the National Health Service...it could create particular problems for healthcare in Northern Ireland, where treatment programs for some rare and serious diseases are designed to work across the entire island of Ireland...“A scenario where the UK leaves without any deal would cause extensive problems for the NHS. It would risk a chaotic disruption to supplies of medical products, and a rise in prices that would push hospitals deeper into deficit,”…Stringent medicine regulations mean manufacturers face multiple Brexit uncertainties, such as the potential need to retest drugs shipped across borders and transfer product licences to different jurisdictions...
- Director of UNLV HIV program kicked off campus (reviewjournal.com)UPDATE: UNLV reopens HIV clinic on day of court hearing (unlvfreepress.com)
The maternal-HIV program at UNLV that was suspended by the university six weeks ago without notice is now without a director...The university...placed Dr. Echezona Ezeanolue, the program’s director, and Dina Patel, a pediatric nurse practitioner, on administrative leave. They were escorted off the campus...Shawn Gerstenberger, dean of the school of community health sciences, asked the university to prosecute Ezeanolue and Patel for various “unsubstantiated claims of wrongdoing.”...the grant-funded program, which provides outpatient HIV primary care services to low-income, vulnerable and medically underserved women, infants, children and youth, was suspended by the university six weeks ago...President Len Jessup said previously that there were irregularities with the way the grant is being administered. Gerstenberger said an administrative audit is underway.
- CMS Shifts Coding and Payment Policy for Biosimilars Under Medicare Part B (raps.org)
The Centers for Medicare and Medicaid Services...announced that it would finalize a policy to separately code and pay for biosimilar products under Medicare Part B, signaling a win for industry...CMS said it is making the change as it was "persuaded that that there is a program need for assigning Part B biosimilar biological products into separate HCPCS [Healthcare Common Procedure Coding System] codes, specifically that this policy change will address concerns about a stronger marketplace, access to these drugs in the United States marketplace, provider and patient choice and competition."...industry groups and companies urged CMS to revise its biosimilar reimbursement policy to provide for separate HCPCS codes for each biosimilar and to reimburse each biosimilar based on its own average sale price..."Effective January 1, 2018, newly approved biosimilar biological products with a common reference product will no longer be grouped into the same HCPCS code. We will issue detailed guidance on coding, including instructions for new codes for biosimilars that are currently grouped into a common payment code and the use of modifiers....