- Pharmacy Week in Review: December 22, 2016 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Senate Aims to Stop Firms From ‘Buying Up Drugs and Jacking Up Prices’ (nytimes.com)
On the heels of headline-grabbing price spikes on prescription drugs, a bipartisan Senate report on Wednesday will call on Congress to take action to prevent huge, unjustified cost increases on decades-old prescription medicines that have no competition...The Senate Special Committee on Aging, reporting the results of a yearlong investigation, said that some drug companies behaved like hedge funds because of the influence of “activist investors.” These companies, the committee said, have developed a “business model that harms patients, taxpayers and the U.S. health care system.”...Our report does not recommend that the government get into the business of setting prices for prescription drugs...We think that would have a harmful impact on the pipeline of innovative drugs...I don’t think we can ignore the market failures that have occurred. The answer is to figure out how we can revitalize the market so that generic drug producers have incentives to compete with companies that are buying up drugs and jacking up prices to make quick, exorbitant profits...
- Looking For Bargains, Many Americans Buy Medicines Abroad (npr.org)
As drug prices have spiraled upward, tens of millions of generally law-abiding Americans have committed an illegal act in response: They have bought prescriptions medicines outside the U.S. and imported them...It's no secret that some Americans regularly buy prescription drugs on the Internet or while traveling abroad. But the popularity of the approach is underscored by the results of a Kaiser Family Foundation poll conducted in November...Eight percent of respondents said they or someone in their household had imported a drug at some point...The Kaiser poll queried a nationally representative sample of 1,202 adults...The Food and Drug Administration has cautioned that many online pharmacies aren't what they seem to be. An international crackdown in 2014 found that many packages of medicines purportedly from Australia, Canada, New Zealand and the U.K. contained drugs from other countries, including India, China and Laos...imported medications could be inferior or expired. Some could be counterfeits. But many medicines purchased from another country are the same as the ones patients buy in the U.S...When purchased outside the country, many prescription medicines cost half or less than they do in the U.S...it is generally illegal for Americans to import drugs into the U.S. for personal use. The law isn't rigorously enforced, in part, because it is difficult to monitor the entry of medicine in suitcases and small packages...in 2015 the FDA implemented a rule that would give government border inspectors expanded authority to destroy drugs imported for personal use at their point of entry.
- NACDS hails rule relaxing access to pharmacy rewards (chaindrugreview.com)
The Department of Health and Human Services is giving pharmacies more flexibility to extend their reward and loyalty programs to beneficiaries in government programs such as Medicare and Medicaid…it will allow government program beneficiaries to enjoy access to programs voluntarily implemented by pharmacies, which reduce health care costs, improve quality and promote patient health...This is a very positive step forward for pharmacy patient care...and of the expanded innovative programs that pharmacies provide...Programs vary in their designs but often include cost savings or other rewards for filling prescriptions or engaging in other health-related activities, such as health screenings, medication adherence programs or healthy lifestyle behaviors…
- Many health news stories lack independent sources (reuters.com)
News stories about medical research rarely include objective comments from experts who weren't involved in the work, according to a recent analysis...Of nearly 600 news articles about medical research published in early 2013, only about one in six included a comment from a person not affiliated with the research - and a quarter of the independent sources quoted in the articles appeared to have no relevant expertise about the topic...commenters frequently had conflicts of interest...As reported in CMAJ (Canadian Medical Association Journal)...researchers identified 131 studies published in major medical journals that were covered by the news media in 591 stories...The average person should be aware that commenters in health new stories quite frequently have (conflicts of interest) that are relevant to the topic at hand, that are frequently not reported and quite likely to influence the disposition of the comments towards the research in question...If we could get more people to look at how we independently and critically vet the evidence as it's presented in news stories, I think high schoolers and maybe even eighth graders can learn to cut through the smoke screen and become better critical thinkers...
- NPAC advocates separation of medical and recreational cannabis (drugstorenews.com)
The Neighbourhood Pharmacy Association of Canada...applauded the federal government's Task Force on Marijuana Legalization and Regulation for recognizing the need to preserve the system of medical cannabis separate from the recreational system...the Task Force clearly noted Neighbourhood Pharmacies' proposition that medical cannabis is a serious drug and that Canadians expect that strength and packaging standards applied to prescription medications be applied to cannabis...We're looking forward to meeting with provincial pharmacy regulatory authorities, the pharmacy community and lawmakers to shape retail rules...We will continue to advocate for patient centric regulations that bring those using medical cannabis into the circle of care…
- What do new FDA letters dinging ‘misleading’ pharma TV ads mean for the industry? (fiercepharma.com)
FDA admonishment of Celgene and Sanofi for DTC TV ads has industry insiders checking the tea leaves for what the letters might signal. The upshot? Possibly an end to overly happy and energetic lifestyle pharma ads...The main complaint in both untitled letters from FDA drug marketing enforcer OPDP (Office of Prescription Drug Promotion) —to Celgene for oral psoriasis treatment Otezla and Sanofi for diabetes drug Toujeo—is distracting music and visuals that interfere with consumers fully understanding the risk information. Both ads have been running since this summer...These commercials are not much different from many other upbeat DTC ads with music...If they are cracking down on these two, then they have decided to look at all of these type of ads. FDA is very subjective about these kind of violations...Sanofi told FiercePharma in an interview last week that it is working to pull the Toujeo TV ads, while Celgene said more broadly that it will work with the FDA to address its concern.
- Nevada shows uptick in enrollments for Affordable Care Act (reviewjournal.com)
About a week after the largest-ever open enrollment day for Affordable Care Act coverage, federal health officials are reporting a year-to-year enrollment increase, with states including Nevada posting an uptick...Nevadans...coverage increased about 3 percent — or by about 1,900 people — over 2015…The number of plan selections...was 60,617 in Nevada, compared with 58,752 by last year’s...deadline...In a statement Wednesday, officials argued the increase showed “clear demand for quality, affordable coverage.”
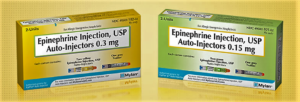
- Mylan launches first generic EpiPen as state AGs sue generics makers for price collusion (drugdeliverybusiness.com)
Mylan launched its 1st generic EpiPen emergency allergy treatment 1 day after attorney generals from 20 states filed a civil complaint against Mylan, Teva Pharmaceuticals, and 4 other generic drug makers. The complaint alleges that the companies co-conspired in informal gatherings, calls and text messages to fix the price for glyburide...and doxycycline hyclate...The generic EpiPen will sell for $300 per two-pack, which is a 50% discount compared to the price of the brand name device...Mylan CEO Heather Bresch said in a statement...“Unfortunately, families will continue to face sticker shock for medications and may be forced to make difficult choices until the pharmaceutical pricing system is reformed to address the increasing shift of costs directly to consumers.”...The 4 other companies targeted by the complaint are Heritage Pharmaceuticals, Aurobindo Pharma, Citron Pharma and Mayne Pharma...
- China’s OxyContin Boom Is a Gold Mine for This Drugmaker (bloomberg.com)
With its harsh anti-narcotics laws and painful history with debilitating opium epidemics in the 19th-century, China wouldn’t spring to mind as a promising market for OxyContin, a painkiller that has been at the center of an opioid addiction outbreak in the U.S…Yet in China, powered by soaring cancer rates and an aging population, OxyContin is turning into a hit. And the drug company behind the brand is giving sales an added boost through an outreach push to physicians and by working with the most powerful of allies—the Chinese government...OxyContin is sold...by Mundipharma (China) Pharmaceutical Co., a company associated with...Purdue Pharma LP, the seller of the long-acting opioid in the U.S...China faces ever greater challenges in managing the use of inherently risky opioids within its sprawling, state-run health-care system.