- Bristol-Myers Squibb to pay $19.5 million to settle off-label promotion case (reuters.com)
Drugmaker Bristol-Myers Squibb Co will pay $19.5 million to resolve multi-state allegations that it improperly promoted a schizophrenia treatment for uses not approved by the U.S. government, New York Attorney General Eric Schneiderman said...The company's agreement with 42 other states and the District of Columbia centers on charges that Bristol-Myers Squibb promoted its Abilify anti-psychotic drug for use in children and elderly patients with dementia and Alzheimer's disease...Besides addressing the off-label promotion allegations, the settlement also resolves charges that the company violated state consumer protection laws by misrepresenting side effects such as metabolic weight gain.
- Following EpiPen controversy, federal watchdog to review Medicaid rebates (statnews.com)
A government watchdog agency is conducting three new reviews of the Medicaid Drug Rebate Program to determine the extent to which drug companies are properly paying required givebacks to the health care program...Under the Medicaid Drug Rebate Program, companies must accurately report — and pay — a rebate on drugs paid for by the agency...The ruckus erupted because Mylan reported EpiPen as a generic product for nearly a decade. This is an important distinction, because classifications are used to determine the size of rebates...are lower for generics — 13 percent versus 23 percent for a brand-name product...The more we learn about the misclassifications, the more we know this program is in dire need of proper oversight...It’s unclear whether CMS took any action between 2009 and now to get the EpiPen classification back on track after being told about the misclassification...
- Pharmacy Week in Review: December 9, 2016 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- The Cost of Counterfeits (pharmtech.com)
The proliferation of counterfeit medicines is nothing new to pharma; however, the scale of the problem seems to be escalating, especially with the Internet providing an easy means for fraudsters to dispense their fakes. Counterfeiting has a devastating impact on public health and the economy. Not only are consumers paying for products of inferior quality, but their well-being is also put at risk. For genuine drugmakers, profits are diluted, but the repercussions extend beyond that...The European Union Intellectual Property Office reported...that the pharmaceutical industry is stripped of approximately €10 billion of revenue every year because of counterfeit medicines; this figure accounts for 4.4% of the sector’s sales...the lost sales translate into 37,700 jobs lost across the pharmaceutical sector in the EU as a result of legitimate manufacturers and distributors employing fewer people than they would do had this problem not existed…With serialization and track-and trace legislations being rolled out over the next few years, pharma is doing its part to secure its supply chain. The problem will be an ongoing challenge for the industry, but with advances in technology, it will become easier to detect the fakes in the near future.
- ‘Bribes,’ ‘kickbacks,’ and ‘fantastic’ nights get ex-Insys CEO, execs indicted for conspiracy (fiercepharma.com)
Prosecutors have worked their way to the top of Insys Therapeutics. After arresting a series of lower-level workers, the Justice Department...hauled in former CEO Michael Babich…Federal authorities alleged that Babich and five other former Insys executives and managers “led a nationwide conspiracy” to bribe doctors to boost scripts for the company’s powerful and addictive painkiller Subsys...indictment...detailed a scheme that Babich and the others allegedly set up with 10 healthcare practitioners in Alabama, Michigan, Texas, Arkansas and elsewhere. The Insys execs pushed sales staff to offer hundreds of thousands of dollars in “bribes and kickbacks” to doctors to increase their Subsys scripts...According to the DOJ, Babich faces charges of conspiracy to commit racketeering, conspiracy to commit wire and mail fraud and conspiracy to violate the Anti-Kickback Law...
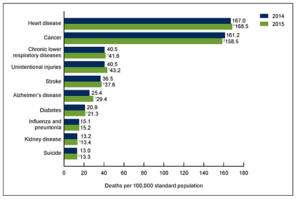
- 5 Findings From the Recent US Mortality Data Report (ajmc.com)The CDC’s National Center for Health Statistics released a report (Mortality in the United States, 2015) with new data on the 2015 US mortality rates. Here are 5 findings to take away from the report...the National Center for Health Statistics...released a data brief containing information on US death rates, life expectancy, and infant mortality rates in 2015. Here are 5 findings to take away from the report.
- In a troubling sign, life expectancy fell.
- The age-adjusted death rate has increased as well.
- The leading causes of death remain the same…
- … But rates of those causes have changed.
- Infant mortality has risen, but not significantly.
- Makers of Abilify drug settle with 43 states, including Nevada, for $19.5M (reviewjournal.com)
Nevada is expected to receive nearly $300,000 in a multistate settlement with a pharmaceutical company accused of marketing a drug to patient groups for which they didn’t have federal approval and mischaracterizing the drug’s risks...Bristol-Myers Squibb Company, which partnered with Otsuka America Pharmaceutical, Inc. to market antipsychotic drug Abilify, settled with 43 states for $19.5 million, according to an announcement Thursday by the office of Nevada Attorney General Adam Laxalt...The company was accused of marketing the drug to seniors with Alzheimer’s disease and dementia despite the fact the drug wasn’t approved for those purposes by the U.S. Food and Drug Administration...
- Supreme Court urged to weigh in on six-month biosimilar delays (fiercepharma.com)
Novartis’ Sandoz unit has been marketing Zarxio, its biosimilar version of Amgen’s blockbuster drug Neupogen, for more than a year, but the drug is still tangled in a patent fight that boils down to one central question: Should biosimilar makers have to give six months' notice to the original drugmaker after they receive FDA approval for their copycat version? The answer will affect not only Novartis and Amgen, but any company seeking to make a biosimilar product…The U.S. Solicitor General...asked the Supreme Court to interpret the 2009 law that originally laid out the proper procedure for seeking FDA approval for biosimilar drugs.
- This Week in Managed Care: December 9, 2016 (ajmc.com)
Sara Belanger, with The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network.
- Five things for pharma marketers to know: Wednesday, December 7, 2016 (mmm-online.com)
- President-elect Donald Trump told Time magazine that he “doesn't like what's happened with drug prices” and that he plans to bring down the cost of prescription medications. Time named Trump its Person of the Year on Wednesday. (Time)
- Mylan plans to lay off nearly 10% of its global workforce. A Mylan spokesperson said the job cuts are part of an efficiency strategy. (BioSpace)
- Pfizer was fined $107 million for overcharging the U.K.'s health system for a generic epilepsy drug, phenytoin sodium. The country's antitrust regulator said that Pfizer deliberately unbranded the drug to skirt a competition law. Pfizer said it plans to appeal the ruling. (WSJ)
- Celgene said its understanding of how its multiple myeloma drug Revlimid works in cancer patients is a boon for its R&D efforts in other diseases. The drugmaker currently has three drugs in its pipeline: CC-122 for diffuse large B-cell lymphoma, CC-220 for lupus, and CC-90009 for acute myeloid leukemia. (Bloomberg)
- ICYMI: GlaxoSmithKline said the CEO of ViiV healthcare, Dr. Dominique Limet, will step down and that Deborah Waterhouse, the company's SVP of primary care at GSK U.S. pharmaceuticals, will succeed him. ViiV Healthcare is an HIV specialty company formed in 2009 by GSK and Pfizer.