- FDA slaps Pfizer’s Hospira unit for ‘misleading’ YouTube ad (fiercepharma.com)
In the first enforcement action from the FDA's marketing police this year, the Office of Prescription Drug Promotion put Hospira in the hot seat over a YouTube video for its sedative Precedex (dexmedetomidine)...The OPDP sent an untitled letter dated Jan. 14 to the Pfizer-owned company, charging the video "omits risks and material facts" about the drug. The agency also rebuked Hospira for publishing the promotional video without submitting it to the OPDP for review...The letter orders Hospira to "cease violating the FD&C Act, as described," and submit a written response before Jan. 29. The response should include a plan for "discontinuing use of such violative materials," the letter states.
- Pharma companies turn to LinkedIn to engage (mmm-online.com)
While the three granddaddy social networks, Facebook, Twitter and YouTube, have spent a decade publicly duking it out for share of human attention-span and commercial usefulness, LinkedIn has quietly and efficiently evolved to become an essential pillar of corporate practice, for individuals and organizations alike...LinkedIn has been something of an enigma and part of the reason has been the difficulty of pinpointing its core purpose. For a long time, it was seen as a professional advancement network, crudely dubbed “Facebook for jobs.” But while career advancement and recruitment is still a major function, over time LinkedIn has evolved into an effective publishing platform for professional content, which has had profound implications for corporate communications and marketing activities...A big part of LinkedIn's appeal lies in the high level of engagement of its members and the potential for precise targeting of content...In the healthcare space, membership encompasses all stakeholder groups: pharma companies, patients, healthcare professionals, marketing agencies, hospitals, investors and, of course, employees...The evolution of LinkedIn...to a publishing platform has had a profound effect on social-media responsibilities within pharma organizations...LinkedIn is now a fully fledged communications vehicle...Regulatory considerations are obviously paramount with any pharma-generated social-media activity...
- Pharma’s December TV spending sags; Pfizer and BMS’ Eliquis rises to top (fiercepharmamarketing.com)
While pharma TV ad spending among the top 10 slowed overall in December, new brands and ongoing promotions still kept the airwaves humming with DTC ads. Spending for the top 10 was $107 million, down from $114 million in November, according to data gathered by real-time TV tracker iSpot.tv....Pfizer and Bristol-Myers Squibb anticoagulant Eliquis rose to the top spot, with the companies plunking down almost $16 million on two different TV spots for the drug...New additions included Pfizer's smoking cessation drug Chantix, which may have been a bid for New Year's resolution-makers, along with Type 2 diabetes meds Toujeo, from Sanofi, and Farxiga, from AstraZeneca. GlaxoSmithKline's respiratory drug Breo Ellipta also joined the top 10 at No. 8...
- Eliquis, Pfizer and Bristol-Myers Squibb anticoagulant, Est. spend: $15.9 million, “Turn Around Your Thinking”
- Prevnar 13, Pfizer pneumococcal pneumonia vaccine, Est. spend: $14.4 million, "One Step"
- Lyrica, Pfizer seizure and pain drug, Est. spend: $14.3 million, “Fibromyalgia Set Backs”
- Humira, AbbVie anti-inflammatory drug, Est. spend: $11.8 million, “Grocery Store”
- Chantix, Pfizer smoking cessation drug, Est. spend: $11.4 million, “Billy”
- Toujeo, Sanofi basal insulin for diabetes, Est. spend: $8.6 million, “Journal”
- Farxiga, AstraZeneca SGLT2 diabetes drug, Est. spend: $8.4 million, “Everyday People”
- Invokana, Johnson & Johnson SGLT2 diabetes drug, Est. spend: $8.2 million, “You're Not Alone”
- Breo Ellipta, GlaxoSmithKline respiratory treatment, $7.5 million, “Missing Piece”
- Xeljanz, Pfizer rheumatoid arthritis drug, $6.6 million, “Made for Better Things”
- French drug trial disaster leaves patient dead amid a slew of unanswered questions (fiercebiotech.com)Scientists call for more details about drug used in deadly French clinical trial (statnews.com)
The patient left brain-dead in last week's botched drug trial has died, French authorities said, spurring more questions about how a routine Phase I study went awry and sent 6 volunteers to the hospital…The patient, dosed with an investigational drug from Portugal's Bial, died Sunday, about a week after the company's CRO halted the study in response to serious side effects. The 5 other hospitalized patients remain in stable condition…In the ensuing fallout, regulators and scientists are struggling to piece together what went wrong in the trial… investigators had yet to determine whether the problems were related to the drug's mechanism of action or the result of contaminated doses…"Together with all the relevant authorities, Bial is strongly committed to ensuring, first of all, the well-being of all participants in this trial and to determine thoroughly and exhaustively the causes which are at the origin of this situation," Bial said in a statement.
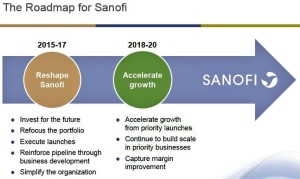
- Sanofi layoffs expected as reorganization goes into effect (statnews.com)
As part of a new "strategic roadmap" unveiled recently, Sanofi plans to announce layoffs that are being described as "sizable" and that will affect a broad swath of company operations in various locations. Some cuts are expected to be...in France, where the drug maker is headquartered, although extracting concessions from French labor unions is notoriously difficult. As a result, a disproportionate number of job losses may occur in the United States. A layoff notice, in any event, is expected to be filed shortly with state officials in New Jersey, where Sanofi maintains a large corporate campus...The drug maker...employs about 110,000 people worldwide, including roughly 17,000 people in the US...Sanofi encountered price discounting in the long-acting insulin market...This was thanks to its Lantus product, which generated about 18 percent of sales in the first half of last year, but faces pressure from payers seeking discounts...a follow-on product is not showing signs of generating needed replacement revenue and a biosimilar version of Lantus will become available at the end of this year. And a deal to sell the MannKind inhaled insulin product known as Afrezza was just ended due to terrible sales.
- Drug industry to fight superbugs together with governments (hosted.ap.org)Pharmaceutical Companies Sign Declaration to Combat Antimicrobial Resistance (pharmtech.com)
Dozens of makers of medicines and diagnostic tests have joined together in an unprecedented effort to tackle "superbugs" - infections that increasingly don't respond to drugs and threaten millions of people in countries rich and poor...74 drugmakers, 11 makers of diagnostic tests and nine industry groups have signed a groundbreaking agreement to work with governments and each other to prevent and improve treatment of drug-resistant infections...have signed the "Declaration on Combating Antimicrobial Resistance."...The declaration calls for steps including:
- governments committing funding to implement the World Health Organization's Global Action Plan to create programs ensuring that health systems use antibiotics appropriately, along with increasing use of fast diagnostic tests and boosting reimbursements for them to ensure patients get the correct treatment.
- better education of doctors and nurses on appropriate antibiotic use.
- improved infection control through better hygiene, vaccination and preventive treatments.
- reduced used of antibiotics in livestock.
- higher reimbursements for antibiotics and diagnostic tests in developed markets.
- more collaboration between researchers at drugmakers and those at universities and government.
- more access to antibiotics in countries around the world.
- Samsung and Biogen win first EU approval for an Enbrel copycat (fiercebiotech.com)Samsung Bioepis Enters the European Biopharmaceutical Market with Benepali®, the First Fusion Protein Biosimilar Approved by the European Commission (finance.yahoo.com)
A joint venture between Biogen and South Korean giant Samsung won Europe's first approval for a lower-cost version of Amgen and Pfizer's blockbuster Enbrel, planning to launch its injection in the coming weeks...The two companies, doing business as Samsung Bioepis, convinced European regulators to clear their Benepali for all of Enbrel's approved indications, including rheumatoid arthritis, psoriatic arthritis, spondyloarthritis and plaque psoriasis…As for Amgen...believes its hold on Enbrel's U.S. rights will keep it safe from a biosimilar challenge in the coming years. In 2011, Amgen secured new patents related to the antibody that the company says will protect the treatment from competition through 2029. Novartis, leading the charge among Enbrel biosimilars developers in the U.S., is hoping to win approval for its version of the treatment this year while mounting a legal challenge on those patents.
- Walgreens CEO Pessina Says Consumer Drug Prices Should Be Lower (bloomberg.com)
Walgreens Boots Alliance Inc. Chief Executive Officer Stefano Pessina said drug prices should be lower for consumers...Costs in the U.S. and other countries are exploding...“It should be lower prices to the end users,”...Pessina, a serial acquirer who built up his British drugstore company Alliance Boots through more than three decades of mergers before selling it to Walgreens, made the comments as drug increases are drawing political scrutiny in the U.S. The House Committee on Oversight and Government Reform is holding a Jan. 26 hearing on the issue, and has asked companies including Valeant Pharmaceuticals International Inc. for details on how they price their products.
- Pfizer looks to Russian partnership to produce some of its meds there, report says (fiercepharma.com)Pfizer to launch joint production with Russia’s Polysan in St. Petersburg — source (tass.ru)
Russia's economy has gotten ugly in the past few years, but that has not dampened the enthusiasm of Big Pharma, which continues to like the look of one of the world's largest emerging markets. Pfizer is reportedly the next up with plans for a partnership to produce some of its meds there...Pfizer is only days away from reporting plans to form a joint venture with Russian drugmaker Polysan to produce Pfizer meds in St. Petersburg...A spokesperson for Polysan would not comment directly but said its policy "is aimed at development of local pharmaceutical industry, including via cooperation with leading global pharma producers."Polysan has its own portfolio of four products that it produces at plants in St. Petersburg and Belgorod, as well as a neurological drug it produces with Germany's Stada...
- Venezuela needs urgent foreign medical aid, pharma group says (reuters.com)
With scores of medicines in short supply due to a severe financial squeeze, Venezuela is suffering a "humanitarian crisis" and requires rapid international assistance, according to a major pharmaceutical association...The Venezuelan Pharmaceutical Federation (Federación Farmacéutica Venezolana) listed 150 medicines, from those for hypertension to cancer, as well as basics such as prophylactics and antibiotics, which are scarce in the OPEC nation of 29 million people..."The national government must accept we are in a humanitarian crisis in the health sector, with patients dying across our territory for lack of medicines," said association president...The health ministry did not respond to a request for comment on the pharmaceutical association's statement, but Venezuela's socialist government has bristled at criticism of its health system...It accuses local groups of exaggerating problems and says the widespread shortages in Venezuela - of food as well as medicines - are due to an "economic war" by foes...Long lines form daily outside pharmacies, and doctors and patients constantly complain of lack of medicines and equipment.