- Maker of generic version of Nexium goes blue to settle litigation (statnews.com)
Dr. Reddy’s Laboratories is feeling blue over a purple pill...The generic drug maker last week agreed to change the color of its new version of the Nexium heartburn medicine from purple to blue in order to settle a lawsuit that was filed by AstraZeneca....For more than two decades, AstraZeneca has capitalized on a marketing campaign that labeled its two widely prescribed heartburn drugs — first, Prilosec and then Nexium — as “the Purple Pill.” But after Dr. Reddy’s last September began selling a lower-cost generic version, which resembled the brand-name drug, AstraZeneca claimed the Indian drug maker was ripping off its trademark look...
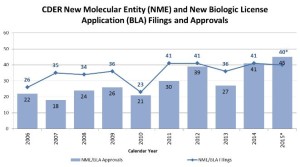
- 2015: Another Strong Year for Patients in Need of New Drug Therapies (blogs.fda.gov)Novel New Drugs Summary 2015 (fda.gov)I’m (John Jenkins,Director of the Office of New Drugs) pleased to report another strong year for FDA approvals of novel new drugs, which offer many patients new treatment options for serious and life-threatening conditions. In 2015, FDA’s Center for Drug Evaluation and Research approved 45 novel new therapies – significantly more than the average of 28 we have approved during the previous nine years of this decade...During this past year, we approved many new drugs to treat various forms of cancer, including four to treat multiple myeloma, and others to treat lung, skin, breast, brain, colorectal, and other cancers. We also approved new drugs to treat heart failure, high cholesterol, cystic fibrosis, and irritable bowel syndrome, as well as the first approved reversal agent for a commonly-used blood thinner...Here are a few highlights of these approvals:
- More than one-third of the novel new drugs CDER approved in 2015 were identified by FDA as “first-in-class,” for example, drugs that use a new and unique mechanism of action for treating a medical condition;
- More than 40% of these new therapies were approved to treat rare or “orphan” diseases that affect 200,000 or fewer Americans–Americans who often have few or no drug treatment options;
- 60% of CDER’s novel new approvals for 2015 were designated in one or more categories of Fast Track, Breakthrough, Priority Review, or Accelerated Approval. Each of these designations helps speed the development and/or approval process and is designed to help bring important medications to the market as quickly as possible; and
- 64% of CDER’s novel new approvals were approved first in the United States before any other country.
- 3 Undiscovered, Undervalued Health Care Stocks Poised to Outperform the Sector in 2016 (thestreet.com)
Small investment ideas can sometimes offer a powerful boost to your portfolio. While they may lack the glitz of the bigger players, these stocks can deliver a strong foundation and long-standing value. Here are three health care stocks that have largely zipped over the heads of Wall Street analysts. They might seem a little riskier in addition to being unknown, but we believe they've got great times ahead of them.
- PetMed Express is a nationwide pet pharmacy. The company markets prescription and nonprescription pet medications and other health products (for dogs, cats, and horses, among other animals) directly to the consumer.
- Meridian Bioscience is an integrated life science entity, involved in the complete diagnostic test kit product development lifecycle. The key applications are for certain strains of viral, respiratory, gastrointestinal and parasitic infectious diseases.
- Amphastar Pharmaceuticals is a specialty pharmaceutical company. It develops, manufactures, markets and sells generic and proprietary injectable and inhalation products. In 2014, the company commenced sales of insulin active pharmaceutical ingredient products.
- Opinion: The AMA is wrong about banning drug ads (statnews.com)
...American Medical Association recently called for a ban on advertising prescription drugs and medical devices directly to consumers. The effort is largely symbolic...But doctors resent the increasing pressure the ads place on them to write prescriptions out of concern patients will switch physicians...they argue that many ads aimed at consumers promote more expensive medicines...and pushes patients to ask for products that either they may not need or is not right for them. This approach is, at best, misguided, and, at worst, ignores the benefits of direct-to-consumer advertising for patients...DTC advertising increases awareness of health problems and leads to a better informed and educated patient who can engage their physician in a dialogue rather than a monologue...So what’s really going on here?...insurers are taking more prescription writing power away from doctors. They first want patients to try generic medications which now make up 88 percent of all available prescription drugs. Second, higher patient copayments for office visits and insurance mean consumers are “shopping” for health care and health care treatments...This makes doctors very uncomfortable. Even with all these changes, research continually validates the notion that patients view their doctors as the gatekeepers to their prescription medicines...DTC advertising leads patients to their health care providers and, depending on the health condition, does not lead to high-priced unnecessary scripts. The AMA should reach out and work with pharma to improve DTC marketing, not request a ban on all DTC ads.
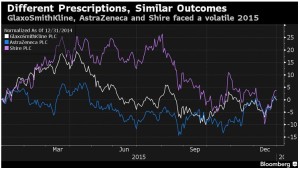
- Will U.K.’s Big 3 Health-Care Stocks Bounce Back in 2016?: Chart (bloomberg.com)
Here’s what lies ahead in 2016 for each company:
- GlaxoSmithKline: CEO Andrew Witty will attend both the J.P. Morgan health-care conference in San Francisco and the World Economic Forum in Davos this month. Shares of the U.K.’s biggest drugmaker have tumbled more than 20 percent since mid-2013. With generics snapping at the heels of its best-selling lung drug Advair, Glaxo has been touting new medicines while shunning large-scale M&A deals.
- AstraZeneca: After spurning a takeover by Pfizer Inc. in 2014, CEO Pascal Soriot has built the company up with small- to mid-sized acquisitions and licensing deals, focusing on drugs for cancer, respiratory disease and diabetes. Generic versions of its blockbuster cholesterol pill Crestor will hit the market in May. Results from several key cancer drug trials are expected in the second half of this year.
- Shire: The smallest of the three companies is said to be in advanced talks with Baxalta Inc. to become the world’s biggest maker of rare-disease drugs. Shares have dropped 21 percent since Aug. 3, the day before its $30 billion offer for Baxalta was made public. On the product front, Shire awaits U.S. approval of a key drug for dry-eye disease called lifitegrast, expected late this year.
- Baxalta in deal to expand immuno-oncology business (reuters.com)
Baxalta Inc has signed a deal with privately held Symphogen under which the companies will develop immuno-oncology drugs to treat rare cancers...The agreement holds a total potential value up to 1.4 billion euros ($1.6 billion) in option fees and milestones over the long term, in addition to royalties on worldwide sales...The deal with Symphogen comes as Baxalta approaches the final stages of negotiating a potential sale to rare diseases drugmaker Shire Pharmaceuticals....Baxalta and Symphogen said...they expect to conduct the first early-stage study in 2017...Immuno-oncology is a new field that uses the method of activating a patient's immune system against tumor growth. Both single and combined immuno-oncology therapies are likely to improve outcomes for a variety of cancers...
- Drug approvals at 19-year high belie industry challenges (reuters.com)
2015 was a good year for innovation in medicine with the Food and Drug Administration approving 45 novel drugs, four more than in 2014 and the most since the all-time record of 53 set in 1996...the European Medicines Agency recommended 93 new products, including generics, up from 82 in 2014...the prospect for further progress in 2016, the pharmaceuticals industry faces challenges, with increased political focus on drug pricing having punctured both biotech and specialty pharma valuations in recent months...The rapid pace of new approvals reflects accelerated review times by regulators, who want to get life-saving treatments to patients, especially in cancer, as well as an improved scientific understanding of diseases...Full drug pipelines at many companies suggest the strong rate of new drug launches is likely to continue for a while yet, with IMS Health forecasting a total of 225 new drug approvals between 2016 and 2020.
- Poor sales prompt Sanofi to pull plug on Mannkind inhaled insulin (reuters.com)
Sanofi is to stop selling an inhalable insulin developed by Mannkind, following disappointing sales of the product since its launch in February 2015...The decision to terminate the collaboration marks a blow for the idea of delivering insulin through an inhaler, rather than by injection...Rights to Afrezza will revert to Mannkind from Sanofi...Mannkind said it was reviewing strategic options for the product, although analysts questioned if the drug had any future...We can't imagine that another legitimate diabetes company would show serious interest in this asset...With little hope for resuscitating Afrezza and a dismal balance sheet (net debt), we see Mannkind in an increasingly precarious position...
- Oregon greenlights pharmacist-prescribed birth control (upi.com)How Oregon Pharmacists Are Prescribing Birth Control (pharmacytimes.com)
Pharmacists in Oregon are now permitted to prescribe birth control pills to qualifying women as part of a wave of new state laws for 2016...Oregon is the first U.S. state to put such a law into effect, with California reportedly looking to follow suit...A doctor's approval is no longer needed for a supply of pills, although experts urge women not to overlook preventative health care in the form of doctor visits...having birth control accessible through a pharmacist doesn't mean preventative health care isn't important...women over age 18 will still be required to fill out a health questionnaire trained pharmacists will use to determine whether to write a prescription. Pharmacists are reportedly still free to refuse prescriptions for religious reasons, but must refer a customer somewhere else.
- WellCare makes CVS Health its pharmacy benefit manager (reuters.com)
Health insurer WellCare Health Plans said it would change its pharmacy benefit manager to CVS Health Corp from UnitedHealth Corp's Optum Rx, effective Jan. 1...About 3.8 million WellCare members enrolled under its Medicaid, Medicare and prescription drug plans will be able to access CVS' pharmacy network, WellCare said on Thursday...CVS is the second-largest U.S. pharmacy benefit manager and drugstore.