- Pharmacy Week in Review: November 20, 2015 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program highlights the latest in pharmacy news, product news, and more. (video)
- FDA approves Adapt Pharma’s nasal spray for opioid overdose treatment (reuters.com)
Food and Drug Administration approved the first-ever nasal spray emergency treatment for opioid overdose…The spray, developed by privately held Adapt Pharma Ltd, uses naloxone, a drug used to treat opioid overdose for nearly 45 years but approved only in injectable forms…The treatment…is expected to have wide coverage under health insurance with affordable co-pays…Group purchasers, such as law enforcement, fire fighters, departments of health, local school districts, colleges and universities, and community-based organizations will be able to purchase the spray at a discounted price of $37.50 per 4 mg device…
- Despite rhetoric, new data shows medicines are not primary driver of premium increases (catalyst.phrma.org)
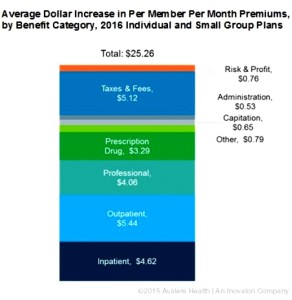
Despite claims from insurers about the impact of medicine costs on premium increases, their own data indicates otherwise. In fact, just $3.29 of the average $25.26 increase in monthly premiums in 2016 is due to prescription drug costs, according to new research released yesterday by Avalere Health. So what is driving premium increases? Avalere found that the largest driver of premium increases was hospital services…analysis is based on actuarially-certified analysis submitted by the plans themselves to justify their premium rate increases. This data shows that prescription drugs dispensed at a pharmacy represent a smaller share of premium increases than inpatient hospitalization, outpatient hospitalization, professional services or taxes and fees. In fact, even if plans did not anticipate any increase in prescription drug spending, the average premium would still increase by more than $20 per month.
- U.S. drug benefit managers clamp down on specialty pharmacies (reuters.com)
...the largest U.S. managers of private prescription drug benefits have cut off at least eight pharmacies that work closely with drugmakers, intensifying scrutiny of a system that helps inflate drug prices…The terminations come from payers who together manage drug benefits for more than 100 million Americans…Express Scripts, the nation's largest pharmacy benefits manager, has changed the algorithms it uses in its audits to find pharmacies focused heavily on one drug manufacturer and has cut ties with half a dozen such pharmacies...Independent pharmacies and their drugmaker partners counter that Express Scripts, CVS and OptumRx together control more than two-thirds of the market through their own mail-order operations. The specialty pharmacies say that the benefit managers are trying to curb the explosive growth of smaller, independent players.
- Post-antibiotic era draws closer as new resistant bacteria discovered (pharmatimes.com)Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study (thelancet.com)
The world is drawing closer to a “post-antibiotic era” after bacteria resistant to last-resort drugs are discovered in China…The report in the Lancet Infectious Diseases identified bacteria resistant to the antibiotic colistin in patients and livestock…the new mutation is able to be easily shared between bacteria…"If this mutation becomes global…and the gene aligns itself with other antibiotic resistance genes…then we will have very likely reached the start of the post-antibiotic era.”… “To advise policy makers on the best way to tackle antimicrobial resistance, robust scientific data on how antimicrobials are used across the EU and how resistance emerges and transfers are needed,”…“This requires close cooperation between the various European agencies that have responsibilities in this area.”
- Price controls loom for generic drugs in India next year (fiercepharmaasia.com)
India's generic drugs could face new price controls as early as next year, as the prime minister's office pushes for an investigation of what one official in press reports called "astronomical" markups by drugmakers, suppliers and retailers…The office reportedly ordered the Department of Pharmaceuticals to look into the large markups that occur on generics that are sold through distributors…a panel already has been established to include the department, the National Pharmaceutical Pricing Authority and the Competition Commission of India, as well as industry groups…For several years, the government has attempted other ways to make generics more affordable to Indians, including the establishment of its own drug store chain, Jan Aushadhi. By the end of the year, the group expects to have more than 400 generics for sale, compared with the 225 now available. Within two years, the government expects to have 3,000 Jan Aushadhi stores throughout the country.
- Global drug spending to hit $1.4 trillion in 2020: IMS (reuters.com)
Global spending on medicines will reach $1.4 trillion in 2020, driven by increased healthcare access in emerging markets and high-priced new drugs for cancer and other diseases…That is up from about $1.07 trillion this year, representing a compound annual growth rate of 4 to 7 percent over the next five years…Some 225 new drugs will come to market over the next five years, about a third aimed at cancer, as well as medicines for rare diseases, which can carry ultra-high price tags, and treatments for autoimmune diseases and heart disease…cost will be partially offset by patent expirations expected to reduce spending on branded medicines by $178 billion, including $41 billion from biologic drugs as cheaper biosimilars become more widely adopted…Developed markets will still account for the lion's share of global spending, about 63 percent, due to higher prices and access to the newest, most expensive treatments.
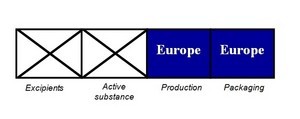
- French proposal for ‘Made in EU’ labels threatens to divide drug industry (statnews.com)Decree on the specification of the place of manufacture on the external packaging of pharmaceutical products (ec.europa.eu)
In a controversial move, France has asked the European Commission to allow new labeling on medicines that would indicate whether products and ingredients come from Europe or elsewhere. But the effort is likely to divide drug makers amid concerns about the quality of prescription drugs…The stated reason for the request is to bolster transparency, because French consumers may have “doubts” about their medicines, according to the filing by the French government. And the notice singled out generic medicines, in particular…The filing also follows the recent suspension of some 700 mostly generic drugs by the European Union. The EU acted after French regulators questioned the reliability of clinical trial data generated by GVK Biosciences, a contract research organization in India that conducts studies for drug makers.
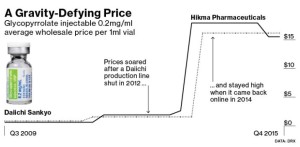
- Broken Market for Old Drugs Means Price Spikes Are Here to Stay (bloomberg.com)
Prices for 50 hospital drugs have risen 10-fold over 8 years…Generic injection meds in short supply as producers drop out…With most products, you’d expect a flood of new supply to quickly drive back down a price spike caused by a temporary shortage. Not so in the topsy-turvy world of hospital pharmaceuticals…All sorts of medications, from treatments for irregular heartbeats to supplies as basic as saline solution, have shot up in price as common hospital drugs are increasingly, and sometimes critically, in short supply. The reasons for the shortages are varied: creaky old factories, an FDA crackdown on quality control, companies exiting the market for more lucrative opportunities and fewer producers in the wake of a wave of mergers and acquisitions…“It’s a broken market," said Stephen Schondelmeyer, a pharmacist and economist at the University of Minnesota who has studied drug prices. "Drug companies know there is going to be an end to this blank check era and they are pushing for whatever they can get."
- American Medical Association wants to ban drug ads to consumers (statnews.com)AMA Calls for Ban on Direct to Consumer Advertising of Prescription Drugs and Medical Devices (ama-assn.org)
American Medical Association is calling for a ban on advertising prescription drugs and medical devices directly to consumers. The move…is largely symbolic, because any ban would have to be authorized by Congress…The new AMA policy comes after years of complaints by physicians…some ads too often encourage patients to seek medicines unnecessarily. They also resent the pressure the ads place on them to write prescriptions out of concern patients will switch physicians…Another rationale for the ban, however, is the rising cost of drugs…many of the ads aimed directly at consumers promote more expensive medicines…the Pharmaceutical Research and Manufacturers of America…“Providing scientifically accurate information to patients so that they are better informed about their health care and treatment options is the goal of direct-to-consumer pharmaceutical advertising...“Beyond increasing patient awareness of disease and available treatments, DTC advertising has been found to…encourages patients to visit their doctors’ offices for important doctor-patient conversations about health that might otherwise not take place.”...