- Pharmacy Podcast – American Pharmacy Purchasing Alliance (APPA) (pharmacypodcast.com)
We interview Kim Kersten – Board of Director member of the American Pharmacy Purchasing Alliance (APPA). (podcast 20:56)
- Novartis, Roche find ‘outcome-based’ drug pricing an elusive dream (reuters.com)
Novartis's heart drug Entresto cuts the risk of re-hospitalization might have helped Chief Executive Joe Jimenez realize his ambition of getting insurers to pay more for treatments when they cut overall medical costs…Instead Jimenez and Severin Schwan, CEO of cross-town rival Roche, have been forced to concede that insurance companies…are not yet ready for such "outcome-based" pricing models…A key hurdle…is that electronic medical record systems aren't capable of accurately tracking a drug's role in reducing hospital stays or preventing further trips to the emergency room…This gap has largely stymied a push to change how drugs are priced and reimbursed by insurers and governments, even though both CEOs contend today's pay-per-pill approach can't be sustained…Drugs account for only around 10 percent of U.S. healthcare costs, Jimenez said, with hospital stays, medical personnel and other costs making up the rest. But existing records systems aren't up to the task of putting this data into perspective.
- Gene therapies offer dramatic promise but shocking costs (washingtonpost.com)
…gene therapy might soon find itself steeped in a new controversy: soaring drug prices…crucial questions about how much patients will pay directly…industry leaders are…talking about ways to get ahead of potentially massive one-time price tags that could make insurers and patients balk…A gene therapy approved in Europe in 2012 costs close to $1 million, and prices are expected to follow suit in the United States. The therapies in the pipeline are mostly for rare genetic diseases: sickle cell, hemophilia or immune deficiency. Their likely high prices stem from the expected value; unlike drugs that a person takes regularly, gene therapies are designed to be given once and have lasting effects…But everyone involved anticipates the potential backlash against a seven-figure price tag, which is leading to radical proposals. Instead of paying for a treatment all at once, insurers and patients could make installment payments as long as the therapy works…Some researchers are adding up the cost of the traditional treatments that a patient will be able to avoid each year to determine a price that, although high, could lead to savings for the health-care system.
- Drug makers kept many clinical trial results a secret: study (pharmalot.com)
When it comes to disclosing clinical trial data, some drug makers are still keeping secrets, according to a new study…Thirty-five percent of all trial results for 15 drugs that were approved in 2012 by the Food and Drug Administration were not publicly disclosed. And nearly 30 percent of the trials conducted for those drugs failed to meet legal disclosure requirements…“This confirms that pharmaceutical companies often fall below legal and ethical standards,”…The findings come amid growing clamor from academics and consumer groups to press drug and device makers to release trial data. If research is not published or reported in accessible registries, physicians and patients are prevented from having a complete picture of the risks and benefits of medicines…Such concerns have been heightened following various safety scandals that revealed trial data for some products was never fully published or disclosed. A few notable examples include…Vioxx.. and…Paxil…Over the past year, regulators in the US and Europe responded to concerns by releasing new rules designed to widen access. The World Health Organization has released a new position statement calling for companies to publish all research studies…Sense About Science, a UK nonprofit that launched the AllTrials campaign to widen access to data, is working with 85 asset managers and pension funds to assess steps taken by drug makers to provide trial information…
- Drug makers beat diabetes lawsuits with an unusual ruling (pharmalot.com)
Several drug makers were handed an unexpected victory this week when a federal judge ruled they weren’t required to update product labels to warn about risks of pancreatic cancer with their diabetes medicines…District Court Judge…explained the labeling changes were not required because the Food and Drug Administration would not have approved those changes. As a result, approximately 750 cases are being tossed…“Right now, it’s a ruling by one district judge in California. So at this point, it doesn’t have huge implications. But there is, potentially, a lot at stake here for the plaintiffs and similar cases,” he tells us. “If the companies were to prevail, it could become an important precedent for companies in similar cases, because it could clarify what a manufacturer has to show to avoid liability.”
- Rite Aid launches genetic testing for Rx (chaindrugreview.com)Theranos isn’t the only diagnostics company exploiting regulatory loopholes (theverge.com)
Rite Aid Corp. has begun offering Harmonyx Diagnostics genetic testing for medications, making it the nation’s first drug chain to provide the service…Rite Aid said…that the self-administered oral swab test — available in all of its pharmacies…helps patients determine the efficacy of their prescription medication based on their genetic makeup…Patients can buy the test kit at any Rite Aid…In the pharmacy, patients then swab inside their cheek, and then a Rite Aid pharmacist takes the sample and returns it to Harmonyx for processing. Harmonyx provides a state-licensed physician to review each patient’s test, and detailed results are sent to the pharmacist and the treating doctor. The results are received as soon as 24 hours after the laboratory receives the test, according to Rite Aid.
- Germany, U.S. in hot pursuit of ‘messenger’ drug molecules (newsdaily.com)
A molecule that carries the recipe for making drugs inside body cells is exciting scientists and investors alike, attracting hundreds of millions of dollars in a scramble for the next promising area of biotechnology… synthetic messenger RNA, or mRNA technology, a new approach to tackling a range of hard-to-treat diseases... In theory, the promise of mRNA is enormous, ranging from cancer to infectious diseases to heart and kidney disorders, since it could be used to tackle the 80 percent of proteins that are difficult to affect with existing medicines…In effect, mRNA serves as software that can be injected into the body to instruct ribosomes, the “3D-printers” found inside cells, to churn out desired proteins…This is a radically different approach from conventional approaches, where therapeutic proteins are produced outside the human body and…then be inserted back into the human body at great complexity and cost…“The field is moving very rapidly,”...“I predict it will have a significant impact.”...
- Why Valeant Is an Unusual Pharmaceutical Ethics Case (bloomberg.com)
Greenhill & Co. CEO Scott Bok discusses the ethics of health care and pharmaceutical companies.
- More than 100 women sue over mispackaged birth control after becoming pregnant (mcall.com)
More than 100 women who became pregnant after allegedly taking mispackaged birthcontrol pills filed suit in Philadelphia last week against Qualitest Inc., a subsidiary of the Irish drug-maker Endo Pharmaceuticals Inc…The case seeks millions in damages, including in some cases the costs of delivering, raising, and educating the children borne of the unplanned pregnancies…The discovery of the mispackaged contraceptives prompted the FDA in 2011 to issue a recall notice for 3.2 million blister packs, according to court filings…. The FDA recall was triggered when a Kansas City woman returned a package to her pharmacist after noticing the blister pack had been rotated 180 degrees, reversing the weekly tablet orientation, according to the suit filed in Philadelphia Court of Common Pleas.
- Drug Prices, Manufacturer Rebates, and the Risk to Channel Economics (drugchannels.net) Prescription Medicines: Costs in Context (phrma.org)
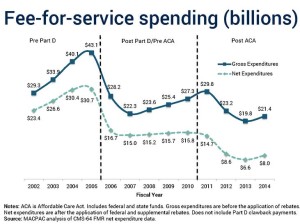
Gross Expenditures = payments to pharmacies Net Expenditures = payments to pharmacies minus federal and state supplemental rebates
When does a price increase not increase the price?...This riddle came to me while reviewing the excellent new Pharmaceutical Research and Manufacturers of America slide deck called Prescription Medicines: Costs in Context. The slides argue—...that pharmaceuticals have been undervalued as a source of health benefits and unfairly maligned as the key driver of costs… Despite the growth in drug list prices, manufacturers are experiencing limited growth in net revenues, which deduct payer rebates. As the data show, this difference has grown sharply over the past two years. Meanwhile, drug list price increases are boosting revenues at drug channels companies—pharmacies, wholesalers, and PBMs. These intermediaries are still compensated on gross (not net) revenues. I wonder: Will the growing gross-to-net disparity make current channel economic arrangements unsustainable for wholesalers and pharmacies?...