- Rise in online pharmacies sees counterfeit drugs go global (thelancet.com)
Increasing public use of online pharmacies, along with a new mass producer of fake medicines, have widened the global market for falsified drugs…. John Clark.. heads up Pfizer's Global Security team…has a question for doctors: do you know where your patients get their drugs from?...The official term for these types of medicines is spurious/falsely-labelled/falsified/counterfeit drugs….developing countries are more than aware of with estimates of the prevalence of counterfeit drugs in some parts of Africa and Asia reaching as high as 70%...it's hard to find a drug that's not being copied or falsified...Counterfeit drugs have been found in 124 countries across all continents and between 2011–14 more than 55 million doses were seized by authorities.
- AmerisourceBergen to buy PharMEDium for $2.58 billion (reuters.com)
Drug distributor AmerisourceBergen Corp agreed to buy PharMEDium Healthcare Holdings Inc for $2.58 billion…expanding its business of supplying compounded drugs to hospitals…"PharMEDium brings ABC a complementary service line that is undergoing a growth renaissance as hospitals more aggressively outsource compounded sterile needs in wake of new government regulations,"…
- Trans-Pacific Partnership Could Reduce Market Exclusivity Period (pharmacytimes.com)TPP negotiators said to reach compromise on biologics (fiercepharmaasia.com)
…Trans-Pacific Partnership negotiations finalized today could affect the cost of health care in significant ways…the United States and its 11 Pacific Rim trade partner countries finalized what will be the largest regional trade accord in history…Negotiations were stalled and then extended due in large part to differences over the appropriate amount of time that pharmaceutical companies should enjoy market exclusivity for a new drug…US negotiators eventually conceded and agreed that brand-name drug manufacturers would have a market exclusivity period of 5 to 8 years…current GPhA president…said the agreement brings the generics industry closer to "improv[ing] worldwide patient access to affordable medicines [by] embracing competition from safe, effective biosimilar therapies."...
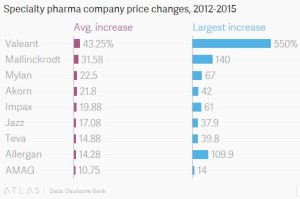
- Massive, unexpected drug price increases are happening all the time (qz.com)
Firms raise prices frequently as they try to make as much money as possible before patents expire, recoup their investments, and make up for the high failure rate of pharmaceutical research…data from Wolters Kluwer’s PriceRx...shows why the implied threat of price controls has pharma investors running so scared. The average price increase for products made by specialty pharmaceutical companies, which concentrate on pricier drugs, was 16% in 2012, 29% in 2013, 22% in 2014, and 19% so far in 2015…Valeant is the biggest standout. …has pursued a strategy of buying out other companies and drugs, slashing R&D budgets, and dramatically increasing prices. Its average price increase so far this year is 66%.
- It’s not easy being green, but biotechs should consider it according to Piramal (in-pharmatechnologist.com)
Pharma and biotech firms not using ‘green chemistry’ to make their products incur higher costs and risk losing market share… There is considerable drug industry interest in ‘green chemistry’ processes that improve efficiency and cut waste – at least that is according to the corporate social responsibility reports issued by firms like Pfizer, GSK and Roche…Whether these efforts are PR – so called greenwashing - or genuine is difficult to judge…while some drugmakers are investing to try and be environmentally-friendly others, particularly smaller biotechs, have yet to commit to greener manufacturing.
- First liquid aspirin’ maker seeks licensing deal (in-pharmatechnologist.com)
Innovate Pharmaceuticals has launched…shelf-stable liquid aspirin, and says it is on the look-out for a licensing partner for the "$500m" market…. Innovate worked with chemicals company Croda for five years on engineering excipients to make what it calls a "truly liquid" aspirin. The pharma firm claims its product allows faster and more complete absorption than other delivery routes, "resulting in potentially drastic reductions in gastric side effects."
- Vetter Embarks on 300-Million Euro Manufacturing Expansion (pharmtech.com)
Vetter will invest approximately 300 million Euros ($335 million) to expand and upgrade its manufacturing facilities...The upgrades are being driven by a changing healthcare market that is affected by issues such as increasingly complex molecules, smaller batch sizes, and more stringent regulatory requirements. Expansion of three sites in Germany will add capacity for logistic services and drug-product manufacturing using an improved system for aseptic processing.
- India considers drug testing labs in each state (fiercepharmaasia.com)
India may house drug testing laboratories in each state under an INR17.5 billion ($270 million) plan to shore up quality… the aim is to have at least one dedicated drug testing laboratory in every state, with 10 states currently not equipped with any testing services for drugs… India approved the spending to upgrade its federal and state drug regulation system with approval from the Cabinet Committee on Economic Affairs…funds, to be spent…on items such as new laboratories and a training academy for regulatory and drug testing officials, are seen as crucial to fixing quality issues that have bedeviled manufacturers and testing firms.
- European API makers must invest to compete with India, says Infa Group (in-pharmatechnologist.com)
Clients are turning their backs on low-cost economies like India and returning to Infa Group, the Italian API maker says…Economies such as India and China have been viewed as low-cost alternatives to Europe and the US for the supply of active pharmaceutical ingredients, but over the past few years, pharma’s changing demands – coupled with concerns over a drop in quality standards – are seeing some customers return back West…This is certainly the case for the Infa Group, one of many European API makers… has seen a number of its main clients return from Asia...
- Drugmaker Tactic to Block Generics May Violate Law, FTC Says (bloomberg.com)
Pharmaceutical companies that make minor tweaks to brand-name drugs in order to blunt competition from cheaper generic treatments may be violating antitrust laws, the Federal Trade Commission said…known as "product-hopping," harms consumers who save billions of dollars each year through generic competition and undermines laws that allow pharmacists to automatically substitute brand-name drugs with low-cost copycats, …"Such conduct could deprive generic companies of their most efficient means of distribution -- automatic substitution at the pharmacy -- and, as a result, maintain the brand’s monopoly through illegal means,"